Yeast biosensor as freeze-dried powders
In order to make our final product as a kit which can be distributed to everyone and be stored for a relatively long time, we use the freeze-drying method to make our engineered yeast as storable powder.
The topic of the effective cell immobilization, long-term storage and subsequent fast recovery to achieve ready-to-use versions of yeast-based systems has been widely studied in the past years. However, while this task has been partially addressed in some industrial processes, e. g. for use in biocatalysts or commercial products, no optimal solutions have been found when applying to biosensors. Nevertheless, most of these strategies significantly diminished the performance of the assays mainly because of affecting sensitivity compared to regular assays. In addition, relevant parameters to long-term storage, namely stability and durability of cells, usually showed lower performances relatively short time after immobilization. While recently, researchers in Czech Republic use freeze-drying method to successfully create a storable yeast biosensor which can maintain its reactivity for 2 month at 4℃ and for 4 month at -18℃[1].
While as the time is limited, we have only completed the freeze-drying process of the constructed yeast glucose sensor (made in stage 1).
After the freeze-drying process, the yeast cells were dried into powders as below:

Fig.1 Freeze-dried samples of yeast constructs
Then we tested the ability of constructed strains reacting to glucose stimulation after different reactivation of freeze-dried powders with medium for 0 or 1 h. We found that, interestingly, the originally promising strain, SMT-pAN failed to respond in a concentration dependent manner, while the strain with milder expression of NLc, which we named SMT-pGN, displayed a delayed glucose concentration-dependent manner after stimulation by glucose PBS solution(Fig.2).
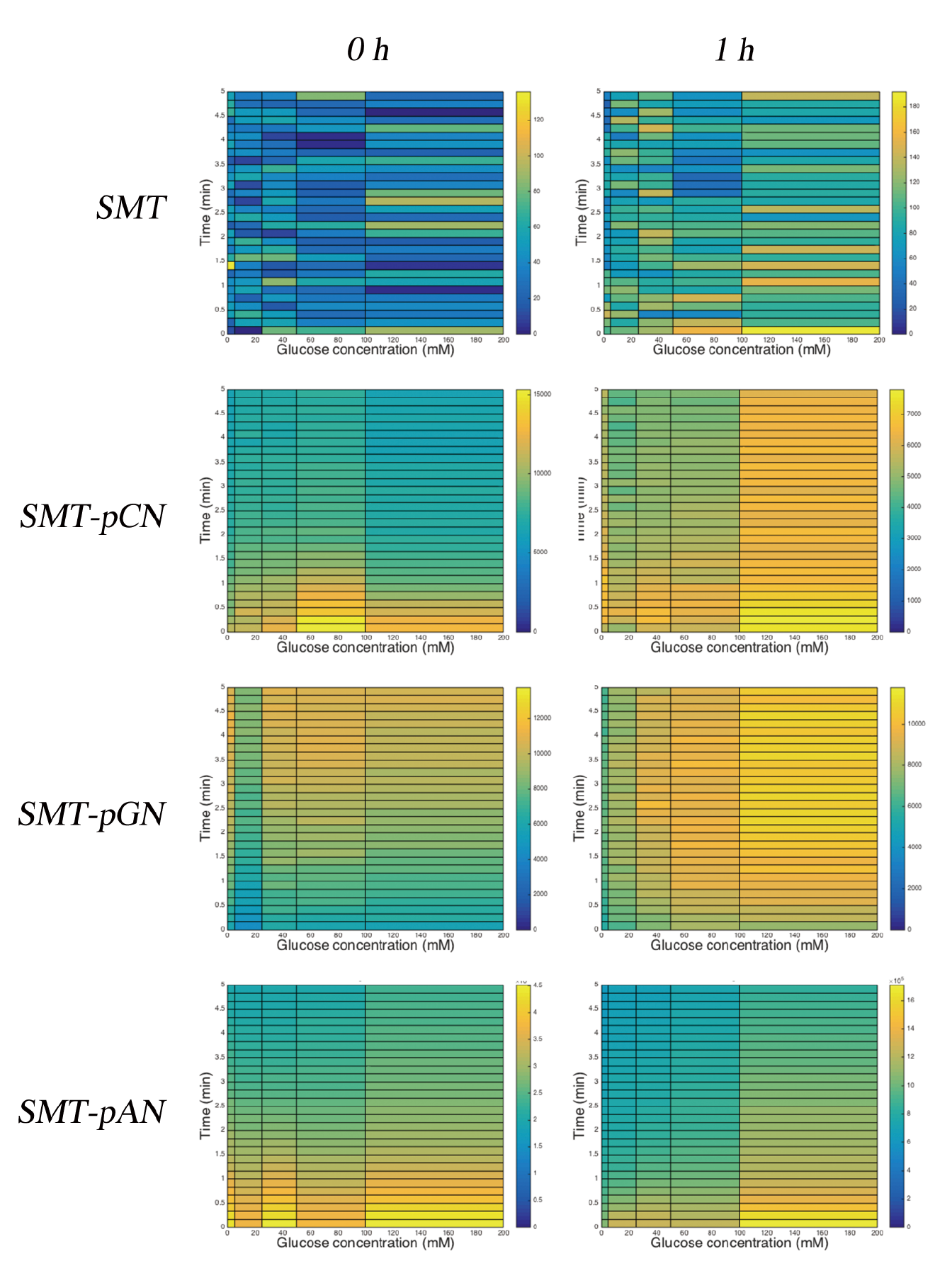
Fig.2 The luminescence response of freeze-dried yeast powders. 0 h and 1 h: After freeze-drying process, yeast powders were put in complex medium in the volume initially used (culture+cryoprotectant) during 30 ℃ for 0 or 1 h for reactivation.
We inferred that this phenomenon was due to the incomplete dissociation of intracellular signaling proteins from each other after the freeze-drying process, as they need space to catalyze and run into one another. Further modifications can be made by setting the reactivation temperature higher (e.g. 30℃) and longer, and most importantly, prepare more parallel groups.
Summary
In this stage, we have:
- 1. Characterize the freeze-drying powders in terms of morphology and methodology..
- 2. Showed the potential of making yeast biosensors active-dry forms for in vitro glucose sensing.
Reference
[1] Jarque, S., M. Bittner, and K. Hilscherova, Freeze-drying as suitable method to achieve ready-to-use yeast biosensors for androgenic and estrogenic compounds. Chemosphere, 2016. 148: p. 204-210.




