(Created page with "{{:Team:SUSTech_Shenzhen/removeStyles}} {{:Team:SUSTech_Shenzhen/themeCss}} {{:Team:SUSTech_Shenzhen/nav}} {{:Team:SUSTech Shenzhen/templates/page-header| url=wiki/images/e/e1...") |
|||
| Line 6: | Line 6: | ||
{{:Team:SUSTech_Shenzhen/main-content-begin}} | {{:Team:SUSTech_Shenzhen/main-content-begin}} | ||
| − | + | = 8 Chromoprotein = | |
| − | + | == Plasmid Construction == | |
ALL ABBREVIATIONS USED: | ALL ABBREVIATIONS USED: | ||
| Line 98: | Line 98: | ||
| 57 | | 57 | ||
|} | |} | ||
| − | + | == 7.22 == | |
| − | + | === Transformation of ①, ③, ⑤ === | |
'''Time:''' 20:30 | '''Time:''' 20:30 | ||
| Line 105: | Line 105: | ||
'''Handler:''' Liao Weiduo | '''Handler:''' Liao Weiduo | ||
| − | + | ==== Procedure: IGEM Protocols ==== | |
1. Thaw competent cells on ice. | 1. Thaw competent cells on ice. | ||
| Line 131: | Line 131: | ||
12. Incubate transformations overnight at 37℃. | 12. Incubate transformations overnight at 37℃. | ||
| − | + | == 7.23 == | |
| − | + | === Picking single colonies of ①, ③, ⑤ === | |
'''Time:''' 16:00 | '''Time:''' 16:00 | ||
| Line 138: | Line 138: | ||
'''Handler:''' Liao Weiduo | '''Handler:''' Liao Weiduo | ||
| − | + | ==== Procedure: ==== | |
# Pick single colonies, adding 6ml LB with corresponding antibiotics, each plate we pick 2 single colonies, A and B. | # Pick single colonies, adding 6ml LB with corresponding antibiotics, each plate we pick 2 single colonies, A and B. | ||
# Incubate overnight at 37℃, 220rpm. | # Incubate overnight at 37℃, 220rpm. | ||
| − | + | == 7.24 == | |
| − | + | === Plasmid extraction of ①, ③, ⑤ A and B === | |
'''Time:''' 9:00 | '''Time:''' 9:00 | ||
| Line 149: | Line 149: | ||
'''Handler:''' Liao Weiduo | '''Handler:''' Liao Weiduo | ||
| − | + | ==== Procedure: bio-tek OMEGA E.Z.N.A. Plasmid DNA Mini Kit I Spin Protocol ==== | |
# Add 2ml LB medium to a 2ml microcentrifuge tube. | # Add 2ml LB medium to a 2ml microcentrifuge tube. | ||
| Line 176: | Line 176: | ||
# Centrifuge at maximum speed for 1 min. | # Centrifuge at maximum speed for 1 min. | ||
| − | + | ==== Results: ==== | |
‘’’Concentration:’’’ ①A: 245.9 ng/ul 1.84; ①B: 340.5 ng/ul 1.84; ③A: 116.2 ng/ul 1.65; ③B: 102.1 ng/ul 1.77; ⑤A: 151.4 ng/ul 1.80; ⑤B: 68.0 ng/ul 1.84; | ‘’’Concentration:’’’ ①A: 245.9 ng/ul 1.84; ①B: 340.5 ng/ul 1.84; ③A: 116.2 ng/ul 1.65; ③B: 102.1 ng/ul 1.77; ⑤A: 151.4 ng/ul 1.80; ⑤B: 68.0 ng/ul 1.84; | ||
| − | + | == 7.25 == | |
| − | + | === Transformation of ②,④,⑥,⑦,⑧,⑨ === | |
'''Time:''' 0:00 | '''Time:''' 0:00 | ||
| Line 187: | Line 187: | ||
'''Handler:''' Tang Shiqiang | '''Handler:''' Tang Shiqiang | ||
| − | + | ==== Procedure: IGEM Protocols ==== | |
| − | + | === Picking single colonies of ②,④,⑥,⑦,⑧,⑨ === | |
'''Time:''' 20:00 | '''Time:''' 20:00 | ||
| Line 195: | Line 195: | ||
'''Handler:''' Tang Shiqiang | '''Handler:''' Tang Shiqiang | ||
| − | + | ==== Procedure: ==== | |
# Pick single colonies, adding 6ml LB with corresponding antibiotics, each plate we pick 2 single colonies, α and β. | # Pick single colonies, adding 6ml LB with corresponding antibiotics, each plate we pick 2 single colonies, α and β. | ||
# Incubate overnight at 37℃, 220rpm. | # Incubate overnight at 37℃, 220rpm. | ||
| − | + | == 7.26 == | |
| − | + | === Plasmid extraction of ②,④,⑥,⑦,⑧,⑨ group α === | |
'''Time:''' 9:40 | '''Time:''' 9:40 | ||
| Line 208: | Line 208: | ||
'''Handler:''' Lu Shixin | '''Handler:''' Lu Shixin | ||
| − | + | ==== Procedure: bio-tek OMEGA E.Z.N.A. Plasmid DNA Mini Kit I Spin Protocol ==== | |
| − | + | ==== Results: ==== | |
Concentration: ②α: 369.8 ng/ul; ④α: 142.4 ng/ul; ⑥α: 116.5 ng/ul; ⑦α: 92 ng/ul; ⑧α: 157.4 ng/ul; ⑨α: 168.3 ng/ul. | Concentration: ②α: 369.8 ng/ul; ④α: 142.4 ng/ul; ⑥α: 116.5 ng/ul; ⑦α: 92 ng/ul; ⑧α: 157.4 ng/ul; ⑨α: 168.3 ng/ul. | ||
| Line 216: | Line 216: | ||
‘’’ Note: ’’’ ⑧ has a red colony. | ‘’’ Note: ’’’ ⑧ has a red colony. | ||
| − | + | === Plasmid extraction of ②,④,⑥,⑦,⑧,⑨ group β === | |
'''Time:''' 14:00 | '''Time:''' 14:00 | ||
| Line 222: | Line 222: | ||
'''Handler:''' Tang Shiqiang | '''Handler:''' Tang Shiqiang | ||
| − | + | ====Procedure: bio-tek OMEGA E.Z.N.A. Plasmid DNA Mini Kit I Spin Protocol ==== | |
| − | + | ==== Results: ==== | |
Concentration: ②β: 496.9 ng/ul 1.86; ④β: 256.4 ng/ul 1.85; ⑥β: 220.3 ng/ul 1.85; ⑦β: 225.7 ng/ul 1.85; ⑧β: 304.6 ng/ul 1.85; ⑨β: 352.2 ng/ul 1.84. | Concentration: ②β: 496.9 ng/ul 1.86; ④β: 256.4 ng/ul 1.85; ⑥β: 220.3 ng/ul 1.85; ⑦β: 225.7 ng/ul 1.85; ⑧β: 304.6 ng/ul 1.85; ⑨β: 352.2 ng/ul 1.84. | ||
| Line 230: | Line 230: | ||
‘’’ Note: ‘’’ ⑧ has a red colony. | ‘’’ Note: ‘’’ ⑧ has a red colony. | ||
| − | + | == 7.27 == | |
| − | + | === Competent Cell Test === | |
'''Time:''' 16:00 | '''Time:''' 16:00 | ||
| Line 238: | Line 238: | ||
'''Handler:''' Lu Shixin | '''Handler:''' Lu Shixin | ||
| − | + | ==== Procedure: IGEM Protocols ==== | |
# Spin down the DNA tubes form the Competent Cell Test Kit to collect all of the DNA into the bottom of each tube prior to use. | # Spin down the DNA tubes form the Competent Cell Test Kit to collect all of the DNA into the bottom of each tube prior to use. | ||
| Line 251: | Line 251: | ||
# Incubate at 37℃ overnight, approximately 16 h. | # Incubate at 37℃ overnight, approximately 16 h. | ||
| − | + | === Enzyme Digestion and Go Gel Electrophoresis Test of ②,④,⑥,⑦,⑧,⑨ === | |
'''Time:''' 20:00 | '''Time:''' 20:00 | ||
| Line 257: | Line 257: | ||
'''Handler:''' Xiao Tianyao | '''Handler:''' Xiao Tianyao | ||
| − | + | ==== Procedure: ==== | |
1. Make enzyme cutting system solution. | 1. Make enzyme cutting system solution. | ||
| Line 400: | Line 400: | ||
<li><p>Go gel electrophoresis.</p></li></ol> | <li><p>Go gel electrophoresis.</p></li></ol> | ||
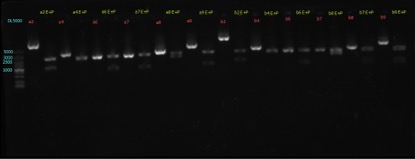
| − | + | ==== Results: ==== | |
figures show below: | figures show below: | ||
| Line 406: | Line 406: | ||
{{SUSTech_Image | filename=SUSTech_Shenzhen-9BCCBDAD-C55F-46B0-B96C-D880F0C7F660.png | caption=‘’’Comment:’’’ ⑧ has some problem, and we will redo it. | width=800px}} | {{SUSTech_Image | filename=SUSTech_Shenzhen-9BCCBDAD-C55F-46B0-B96C-D880F0C7F660.png | caption=‘’’Comment:’’’ ⑧ has some problem, and we will redo it. | width=800px}} | ||
| − | + | == 7.28 == | |
| − | + | === Competent Cell Test Colonies Counting === | |
'''Time:''' 13:00 | '''Time:''' 13:00 | ||
| Line 414: | Line 414: | ||
'''Handler:''' Lu Shixin | '''Handler:''' Lu Shixin | ||
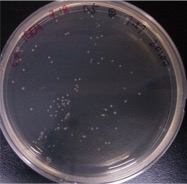
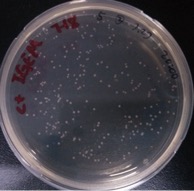
| − | + | ==== Result: ==== | |
{{SUSTech_Image | filename=SUSTech_Shenzhen-C89646A9-AD79-4481-95BB-E54BBC7C1B87.png | caption=Figure 1. Water control | width=300px}} | {{SUSTech_Image | filename=SUSTech_Shenzhen-C89646A9-AD79-4481-95BB-E54BBC7C1B87.png | caption=Figure 1. Water control | width=300px}} | ||
{{SUSTech_Image | filename=SUSTech_Shenzhen-D6D86280-E0BE-43CE-A096-8C835F4D8426.png | caption= Figure 2. 0.5 pg/ul | width=300px}} | {{SUSTech_Image | filename=SUSTech_Shenzhen-D6D86280-E0BE-43CE-A096-8C835F4D8426.png | caption= Figure 2. 0.5 pg/ul | width=300px}} | ||
| Line 422: | Line 422: | ||
And all the colonies are red under UV light. | And all the colonies are red under UV light. | ||
| − | + | == 7.29 == | |
| − | + | === Transformation of ⑧,⑩,11 === | |
'''Time:''' 16:00 | '''Time:''' 16:00 | ||
| Line 430: | Line 430: | ||
'''Handler:''' Lu Shixin, Tang Shiqiang | '''Handler:''' Lu Shixin, Tang Shiqiang | ||
| − | + | ==== Procedure: IGEM Transformation Protocol ==== | |
| − | + | === Enzyme Digestion for Gel Extraction of ⑤,⑥,⑦,⑧,⑨ === | |
'''Time:''' 20:00 | '''Time:''' 20:00 | ||
| Line 438: | Line 438: | ||
'''Handler:''' LuShixin, Tang Shiqiang | '''Handler:''' LuShixin, Tang Shiqiang | ||
| − | + | ==== Procedure: ==== | |
# Make enzyme cutting system solution. | # Make enzyme cutting system solution. | ||
| Line 519: | Line 519: | ||
# Incubate at 37℃ overnight. (Start from 21:20) | # Incubate at 37℃ overnight. (Start from 21:20) | ||
| − | + | == 7.30 == | |
| − | + | === Picking single colonies of ⑧, ⑩, 11 === | |
'''Time:''' 9:00 | '''Time:''' 9:00 | ||
| Line 527: | Line 527: | ||
'''Handler:''' Lu Shixin | '''Handler:''' Lu Shixin | ||
| − | + | ==== Procedure: ==== | |
# Pick single colonies, adding 1ml LB with corresponding antibiotics in 1.5ml EP tube, each plate we pick 2 single colonies, α and β. | # Pick single colonies, adding 1ml LB with corresponding antibiotics in 1.5ml EP tube, each plate we pick 2 single colonies, α and β. | ||
| Line 534: | Line 534: | ||
# Incubate at 37℃ for 10h, 220rpm. | # Incubate at 37℃ for 10h, 220rpm. | ||
| − | + | === Go Gel Electrophoresis and Gel Extraction of ⑤,⑥,⑦,⑧,⑨ === | |
'''Time:''' 9:00 | '''Time:''' 9:00 | ||
| Line 540: | Line 540: | ||
'''Handler:''' Lu Shixin | '''Handler:''' Lu Shixin | ||
| − | + | ==== Procedure: bio-tek OMEGA E.Z.N.A. Gel Extraction Kit – Spin Protocol ==== | |
# Make 1% agarose gel 30ml. | # Make 1% agarose gel 30ml. | ||
| Line 567: | Line 567: | ||
# Centrifuge at maximum speed for 1 min. | # Centrifuge at maximum speed for 1 min. | ||
| − | + | ==== Results: ==== | |
{{SUSTech_Image | filename=SUSTech_Shenzhen-4E6C74F1-3B4C-4F1A-B283-6751068590D8.png | caption= | width=500px}} | {{SUSTech_Image | filename=SUSTech_Shenzhen-4E6C74F1-3B4C-4F1A-B283-6751068590D8.png | caption= | width=500px}} | ||
| Line 573: | Line 573: | ||
All the concentration of gel extraction are below 10 ng/ul. So we decide to repeat enzyme digestion with more DNA. | All the concentration of gel extraction are below 10 ng/ul. So we decide to repeat enzyme digestion with more DNA. | ||
| − | + | === Enzyme Digestion for Gel Extraction of ⑤,⑥,⑦,⑨ (Redo) === | |
'''Time:''' 18:00 | '''Time:''' 18:00 | ||
| Line 579: | Line 579: | ||
'''Handler:''' Lu Shixin | '''Handler:''' Lu Shixin | ||
| − | + | ==== Procedure: ==== | |
# Make enzyme cutting system solution. | # Make enzyme cutting system solution. | ||
| Line 647: | Line 647: | ||
# Incubate at 37℃ for 3h. (Start from 18:40) | # Incubate at 37℃ for 3h. (Start from 18:40) | ||
| − | + | === Plasmid extraction of ⑧,⑩,11 === | |
'''Time:''' 21:00 | '''Time:''' 21:00 | ||
| Line 653: | Line 653: | ||
'''Handler:''' Lu Shixin | '''Handler:''' Lu Shixin | ||
| − | + | ==== Procedure:''' '''bio-tek OMEGA E.Z.N.A. Plasmid DNA Mini Kit I Spin Protocol ==== | |
| − | + | ==== Results: ==== | |
The length of them are correct. | The length of them are correct. | ||
| Line 661: | Line 661: | ||
Concentration: ⑧: 130.2 ng/ul; ⑩: 147.0 ng/ul; 11: 93.3 ng/ul. | Concentration: ⑧: 130.2 ng/ul; ⑩: 147.0 ng/ul; 11: 93.3 ng/ul. | ||
| − | + | === Enzyme Digestion for Gel Extraction of ⑧,⑩,11 === | |
'''Time:''' 22:30 | '''Time:''' 22:30 | ||
| Line 667: | Line 667: | ||
'''Handler:''' Lu Shixin | '''Handler:''' Lu Shixin | ||
| − | + | ==== Procedure: ==== | |
# Make enzyme cutting system solution. | # Make enzyme cutting system solution. | ||
| Line 726: | Line 726: | ||
# Incubate at 37℃ overnight. (Start from 22:40) | # Incubate at 37℃ overnight. (Start from 22:40) | ||
| − | + | === Go Gel Electrophoresis and Gel Extraction of ⑤,⑥,⑦,⑨ (Redo) === | |
'''Time:''' 22:00 | '''Time:''' 22:00 | ||
| Line 732: | Line 732: | ||
'''Handler:''' Tang Shiqiang | '''Handler:''' Tang Shiqiang | ||
| − | + | ==== Procedure: bio-tek OMEGA E.Z.N.A. Gel Extraction Kit – Spin Protocol ==== | |
| − | + | ==== Results: ==== | |
Gel extraction concentration: ⑤: 15.3 ng/ul; ⑥: 36.3 ng/ul; ⑦: 36.8 ng/ul; ⑨: 26.9 ng/ul. | Gel extraction concentration: ⑤: 15.3 ng/ul; ⑥: 36.3 ng/ul; ⑦: 36.8 ng/ul; ⑨: 26.9 ng/ul. | ||
| − | + | == 7.31 == | |
| − | + | === Ligation of ⑥+⑤, ⑦+⑤, ⑨+⑤ === | |
'''Time:''' 0:00 | '''Time:''' 0:00 | ||
| Line 746: | Line 746: | ||
'''Handler:''' Tang Shiqiang | '''Handler:''' Tang Shiqiang | ||
| − | + | ==== Procedure: ==== | |
# Make ligation system solution. | # Make ligation system solution. | ||
| Line 790: | Line 790: | ||
# Take 2.5ul ligation system solution to do transformation. | # Take 2.5ul ligation system solution to do transformation. | ||
| − | + | === Go Gel Electrophoresis and Gel Extraction of ⑧, ⑩, 11 === | |
'''Time:''' 15:30 | '''Time:''' 15:30 | ||
| Line 796: | Line 796: | ||
'''Handler:''' Lu Shixin | '''Handler:''' Lu Shixin | ||
| − | + | ==== Procedure: bio-tek OMEGA E.Z.N.A. Gel Extraction Kit – Spin Protocol ==== | |
| − | + | ==== Results: ==== | |
{{SUSTech_Image | filename=SUSTech_Shenzhen-FF23D23B-B68F-42EB-87A2-DBDED38BDF8E.png | caption= | width=500px}} | {{SUSTech_Image | filename=SUSTech_Shenzhen-FF23D23B-B68F-42EB-87A2-DBDED38BDF8E.png | caption= | width=500px}} | ||
| Line 806: | Line 806: | ||
‘’‘Note:’‘’ We found that we take a wrong ⑧ before. | ‘’‘Note:’‘’ We found that we take a wrong ⑧ before. | ||
| − | + | === Transformation of ⑧ === | |
'''Time:''' 16:30 | '''Time:''' 16:30 | ||
| Line 812: | Line 812: | ||
'''Handler:''' Tang Shiqiang | '''Handler:''' Tang Shiqiang | ||
| − | + | ==== Procedure: IGEM Transformation Protocol ==== | |
| − | + | === Picking single colonies of ⑥+⑤, ⑦+⑤, ⑨+⑤ === | |
'''Time:''' 19:30 | '''Time:''' 19:30 | ||
| Line 820: | Line 820: | ||
'''Handler:''' Tang Shiqiang | '''Handler:''' Tang Shiqiang | ||
| − | + | ==== Procedure: ==== | |
# Pick single colonies, adding 6ml LB with corresponding antibiotics, each plate we pick 2 single colonies, A and B. | # Pick single colonies, adding 6ml LB with corresponding antibiotics, each plate we pick 2 single colonies, A and B. | ||
# Incubate overnight at 37℃, 220rpm. | # Incubate overnight at 37℃, 220rpm. | ||
| − | + | == 8.1 == | |
| − | + | === Picking single colonies of ⑧ === | |
'''Time:''' 9:30 | '''Time:''' 9:30 | ||
| Line 833: | Line 833: | ||
'''Handler:''' Tang Shiqiang | '''Handler:''' Tang Shiqiang | ||
| − | + | ==== Procedure: ==== | |
# Pick single colonies, adding 6ml LB with corresponding antibiotics, each plate we pick 2 single colonies, A and B. | # Pick single colonies, adding 6ml LB with corresponding antibiotics, each plate we pick 2 single colonies, A and B. | ||
# Incubate at 37℃ for about 14h, 220rpm. | # Incubate at 37℃ for about 14h, 220rpm. | ||
| − | + | === Plasmid extraction of ⑥+⑤, ⑦+⑤, ⑨+⑤ A and B === | |
'''Time:''' 9:30 | '''Time:''' 9:30 | ||
| Line 844: | Line 844: | ||
'''Handler:''' Tang Shiqiang | '''Handler:''' Tang Shiqiang | ||
| − | + | ==== Procedure: bio-tek OMEGA E.Z.N.A. Plasmid DNA Mini Kit I Spin Protocol ==== | |
| − | + | ==== Results: ==== | |
Concentration: ⑥+⑤A: 126.3 ng/ul 1.86; ⑥+⑤B: 124.9 ng/ul 1.82; ⑦+⑤A: 116.5 ng/ul 1.87; ⑦+⑤B: 112.0 ng/ul 1.86; ⑨+⑤A: 120.4 ng/ul 1.87; ⑨+⑤B: 149.0 ng/ul 1.87; | Concentration: ⑥+⑤A: 126.3 ng/ul 1.86; ⑥+⑤B: 124.9 ng/ul 1.82; ⑦+⑤A: 116.5 ng/ul 1.87; ⑦+⑤B: 112.0 ng/ul 1.86; ⑨+⑤A: 120.4 ng/ul 1.87; ⑨+⑤B: 149.0 ng/ul 1.87; | ||
| − | + | === Enzyme Digestion and Go Gel Electrophoresis Test of ⑥+⑤, ⑦+⑤, ⑨+⑤ A and B === | |
'''Time:''' 12:00 | '''Time:''' 12:00 | ||
| Line 856: | Line 856: | ||
'''Handler:''' Tang Shiqiang | '''Handler:''' Tang Shiqiang | ||
| − | + | ==== Procedure: ==== | |
# Make enzyme cutting system solution. | # Make enzyme cutting system solution. | ||
| Line 924: | Line 924: | ||
# Go gel electrophoresis. | # Go gel electrophoresis. | ||
| − | + | ==== Results: ==== | |
{{SUSTech_Image | filename=SUSTech_Shenzhen-3DD57F02-827C-43BC-B57F-2ACCA230ECF4.png | caption= | width=500px}} | {{SUSTech_Image | filename=SUSTech_Shenzhen-3DD57F02-827C-43BC-B57F-2ACCA230ECF4.png | caption= | width=500px}} | ||
| Line 931: | Line 931: | ||
‘’‘Note:’‘’ Finally we found that we used a tube of wrong ⑤. | ‘’‘Note:’‘’ Finally we found that we used a tube of wrong ⑤. | ||
| − | + | === Put ⑧ A and B at 4℃ === | |
'''Time:''' 20:00 | '''Time:''' 20:00 | ||
| Line 937: | Line 937: | ||
'''Handler:''' Tang Shiqiang | '''Handler:''' Tang Shiqiang | ||
| − | + | == 8.2 == | |
| − | + | === Plasmid extraction of ⑧ A and B === | |
'''Time:''' 14:00 | '''Time:''' 14:00 | ||
| Line 945: | Line 945: | ||
'''Handler:''' Tang Shiqiang | '''Handler:''' Tang Shiqiang | ||
| − | + | ==== Procedure: bio-tek OMEGA E.Z.N.A. Plasmid DNA Mini Kit I Spin Protocol ==== | |
| − | + | ==== Results: ==== | |
Concentration: ⑧A: 116.5 ng/ul 1.82; ⑧B: 73.2 ng/ul 1.81. | Concentration: ⑧A: 116.5 ng/ul 1.82; ⑧B: 73.2 ng/ul 1.81. | ||
| − | + | === Enzyme Digestion for Gel Extraction of ⑤, ⑧ === | |
'''Time:''' 22:00 | '''Time:''' 22:00 | ||
| Line 957: | Line 957: | ||
'''Handler:''' Lu Shixin, Tang Shiqiang | '''Handler:''' Lu Shixin, Tang Shiqiang | ||
| − | + | ==== Procedure: ==== | |
# Make enzyme cutting system solution. | # Make enzyme cutting system solution. | ||
| Line 1,011: | Line 1,011: | ||
# Incubate at 37℃ overnight. (Start from 22:25) | # Incubate at 37℃ overnight. (Start from 22:25) | ||
| − | + | == 8.3 == | |
| − | + | === Go Gel Electrophoresis and Gel Extraction of ⑤, ⑧ === | |
'''Time:''' 10:00 | '''Time:''' 10:00 | ||
| Line 1,019: | Line 1,019: | ||
'''Handler:''' Lu Shixin | '''Handler:''' Lu Shixin | ||
| − | + | ==== Procedure: bio-tek OMEGA E.Z.N.A. Gel Extraction Kit – Spin Protocol ==== | |
| − | + | ==== Results: ==== | |
{{SUSTech_Image | filename=SUSTech_Shenzhen-0D26F48D-11BF-4C72-B460-51AB3110125F.png | caption= | width=500px}} | {{SUSTech_Image | filename=SUSTech_Shenzhen-0D26F48D-11BF-4C72-B460-51AB3110125F.png | caption= | width=500px}} | ||
Concentration: ⑤: 51.4 ng/ul 1.75; ⑧: 23.2 ng/ul 1.71. | Concentration: ⑤: 51.4 ng/ul 1.75; ⑧: 23.2 ng/ul 1.71. | ||
| − | + | === Ligation of ⑥+⑤, ⑦+⑤, ⑧+⑤, ⑨+⑤ === | |
'''Time:''' 16:30 | '''Time:''' 16:30 | ||
| Line 1,032: | Line 1,032: | ||
'''Handler:''' Lu Shixin | '''Handler:''' Lu Shixin | ||
| − | + | ==== Procedure: ==== | |
# Make ligation system solution. | # Make ligation system solution. | ||
| Line 1,084: | Line 1,084: | ||
# Take 2ul ligation system solution to do transformation. | # Take 2ul ligation system solution to do transformation. | ||
| − | + | == 8.4 == | |
| − | + | === Picking single colonies of ⑥+⑤, ⑦+⑤, ⑧+⑤, ⑨+⑤ === | |
'''Time:''' 9:30 | '''Time:''' 9:30 | ||
| Line 1,092: | Line 1,092: | ||
'''Handler:''' Lu Shixin | '''Handler:''' Lu Shixin | ||
| − | + | ==== Procedure: ==== | |
# Pick single colonies, adding 6ml LB with corresponding antibiotics, each plate we pick 2 single colonies, A and B. | # Pick single colonies, adding 6ml LB with corresponding antibiotics, each plate we pick 2 single colonies, A and B. | ||
# Incubate at 37℃ for about 12h, 220rpm. | # Incubate at 37℃ for about 12h, 220rpm. | ||
| − | + | === Plasmid extraction of ⑥+⑤, ⑦+⑤, ⑧+⑤, ⑨+⑤ A and B === | |
'''Time:''' 21:30 | '''Time:''' 21:30 | ||
| Line 1,103: | Line 1,103: | ||
'''Handler:''' Tang Shiqiang | '''Handler:''' Tang Shiqiang | ||
| − | + | ==== Procedure: bio-tek OMEGA E.Z.N.A. Plasmid DNA Mini Kit I Spin Protocol ==== | |
| − | + | ==== Results: ==== | |
Concentration: ⑥+⑤A: 54.4 ng/ul 2.18; ⑥+⑤B: 29.2 ng/ul 3.15; ⑦+⑤A: 55.4 ng/ul 2.42; ⑦+⑤B: 58.8 ng/ul 2.42; ⑧+⑤A: 70.0 ng/ul 2.22; ⑧+⑤B: 35.6 ng/ul 2.84; ⑨+⑤A: 65.9 ng/ul 2.23; ⑨+⑤B: 74.3 ng/ul 2.18; | Concentration: ⑥+⑤A: 54.4 ng/ul 2.18; ⑥+⑤B: 29.2 ng/ul 3.15; ⑦+⑤A: 55.4 ng/ul 2.42; ⑦+⑤B: 58.8 ng/ul 2.42; ⑧+⑤A: 70.0 ng/ul 2.22; ⑧+⑤B: 35.6 ng/ul 2.84; ⑨+⑤A: 65.9 ng/ul 2.23; ⑨+⑤B: 74.3 ng/ul 2.18; | ||
| Line 1,111: | Line 1,111: | ||
‘’’Comments:’’’ All the concentration seems to have some problem, and the A260/280 have some problem. | ‘’’Comments:’’’ All the concentration seems to have some problem, and the A260/280 have some problem. | ||
| − | + | === Enzyme Digestion and Go Gel Electrophoresis Test of ⑥+⑤, ⑦+⑤, ⑧+⑤, ⑨+⑤ A and B === | |
'''Time:''' 23:00 | '''Time:''' 23:00 | ||
| Line 1,117: | Line 1,117: | ||
'''Handler:''' Tang Shiqiang | '''Handler:''' Tang Shiqiang | ||
| − | + | ==== Procedure: ==== | |
# Make enzyme cutting system solution. | # Make enzyme cutting system solution. | ||
| Line 1,199: | Line 1,199: | ||
# Go gel electrophoresis. | # Go gel electrophoresis. | ||
| − | + | ==== Results: ==== | |
The length seems to be correct. | The length seems to be correct. | ||
| − | + | == 8.5 == | |
| − | + | === Enzyme Digestion for Gel Extraction of ⑥+⑤, ⑦+⑤, ⑧+⑤, ⑨+⑤ === | |
'''Time:''' 2:20 | '''Time:''' 2:20 | ||
| Line 1,211: | Line 1,211: | ||
'''Handler:''' Tang Shiqiang | '''Handler:''' Tang Shiqiang | ||
| − | + | ==== Procedure: ==== | |
# Make enzyme cutting system solution. | # Make enzyme cutting system solution. | ||
| Line 1,263: | Line 1,263: | ||
<li><p>Incubate at 37℃ overnight. (Start from 2:30)</p></li></ol> | <li><p>Incubate at 37℃ overnight. (Start from 2:30)</p></li></ol> | ||
| − | + | === Go Gel Electrophoresis and Gel Extraction of ⑥+⑤, ⑦+⑤, ⑧+⑤, ⑨+⑤ === | |
'''Time:''' 8:10 | '''Time:''' 8:10 | ||
| Line 1,269: | Line 1,269: | ||
'''Handler:''' Lu Shixin, Tang Shiqiang | '''Handler:''' Lu Shixin, Tang Shiqiang | ||
| − | + | ==== Procedure: bio-tek OMEGA E.Z.N.A. Gel Extraction Kit – Spin Protocol ==== | |
| − | + | ==== Results: ==== | |
Concentration: ⑥+⑤: 25.1 ng/ul 1.58; ⑦+⑤: 23.4 ng/ul 1.54; ⑧+⑤: 45.9 ng/ul 1.01; ⑨+⑤: 26.7 ng/ul 1.50; | Concentration: ⑥+⑤: 25.1 ng/ul 1.58; ⑦+⑤: 23.4 ng/ul 1.54; ⑧+⑤: 45.9 ng/ul 1.01; ⑨+⑤: 26.7 ng/ul 1.50; | ||
| − | + | === Ligation of ⑩,11 and ⑥+⑤, ⑦+⑤, ⑧+⑤, ⑨+⑤ === | |
'''Time:''' 11:00 | '''Time:''' 11:00 | ||
| Line 1,281: | Line 1,281: | ||
'''Handler:''' Lu Shixin, Tang Shiqiang | '''Handler:''' Lu Shixin, Tang Shiqiang | ||
| − | + | ==== Procedure: ==== | |
# Make ligation system solution. | # Make ligation system solution. | ||
| Line 1,378: | Line 1,378: | ||
# Take 5ul ligation system solution to do transformation. | # Take 5ul ligation system solution to do transformation. | ||
| − | + | == 8.6 == | |
| − | + | === Observation of single colonies of ⑩+⑥+⑤, ⑩+⑦+⑤, ⑩+⑧+⑤, ⑩+⑨+⑤, 11+⑥+⑤, 11+⑦+⑤, 11+⑧+⑤, 11+⑨+⑤ === | |
'''Time:''' 7:00 | '''Time:''' 7:00 | ||
| Line 1,386: | Line 1,386: | ||
'''Handler:''' Xiao Tianyao | '''Handler:''' Xiao Tianyao | ||
| − | + | ==== Results: ==== | |
{| class=" table table-striped" | {| class=" table table-striped" | ||
| Line 1,425: | Line 1,425: | ||
Comments: Control groups have colonies. So the ligation results are improbable. We would redo it. | Comments: Control groups have colonies. So the ligation results are improbable. We would redo it. | ||
| − | + | === Picking single colonies of ⑩+⑦+⑤, ⑩+⑧+⑤, ⑩+⑨+⑤, 11+⑥+⑤, 11+⑦+⑤, 11+⑧+⑤, 11+⑨+⑤ === | |
'''Time:''' 10:00 | '''Time:''' 10:00 | ||
| Line 1,431: | Line 1,431: | ||
'''Handler:''' Liao Weiduo | '''Handler:''' Liao Weiduo | ||
| − | + | ==== Procedure: ==== | |
# Pick single colonies, adding 3 ml C+ LB. | # Pick single colonies, adding 3 ml C+ LB. | ||
# Incubate at 37℃ for about 12h, 220rpm. | # Incubate at 37℃ for about 12h, 220rpm. | ||
| − | + | === Picking single colonies of ⑥+⑤, ⑦+⑤, ⑧+⑤, ⑨+⑤ === | |
'''Time:''' 11:00 | '''Time:''' 11:00 | ||
| Line 1,442: | Line 1,442: | ||
'''Handler:''' Tang Shiqiang | '''Handler:''' Tang Shiqiang | ||
| − | + | ==== Procedure: ==== | |
# Pick single colonies, adding 6ml LB with corresponding antibiotics, each plate we pick 2 single colonies, C and D. | # Pick single colonies, adding 6ml LB with corresponding antibiotics, each plate we pick 2 single colonies, C and D. | ||
# Incubate at 37℃ for about 12h, 220rpm. | # Incubate at 37℃ for about 12h, 220rpm. | ||
| − | + | === Plasmid extraction of ⑥+⑤, ⑦+⑤, ⑧+⑤, ⑨+⑤ A and B === | |
'''Time:''' 23:00 | '''Time:''' 23:00 | ||
| Line 1,453: | Line 1,453: | ||
'''Handler:''' Tang Shiqiang | '''Handler:''' Tang Shiqiang | ||
| − | + | ==== Procedure: bio-tek OMEGA E.Z.N.A. Plasmid DNA Mini Kit I Spin Protocol ==== | |
| − | + | ==== Results: ==== | |
Concentration: ⑥+⑤C: 63.8 ng/ul 1.81; ⑥+⑤D: 59.4 ng/ul 1.79; ⑦+⑤C: 49.9 ng/ul 1.81; ⑦+⑤D: 65.8 ng/ul 1.75; ⑧+⑤C: 73.4ng/ul 1.81; ⑧+⑤D: 72.8 ng/ul 1.81; ⑨+⑤C: 65.8 ng/ul 1.74; ⑨+⑤D: 64.1 ng/ul 1.83. | Concentration: ⑥+⑤C: 63.8 ng/ul 1.81; ⑥+⑤D: 59.4 ng/ul 1.79; ⑦+⑤C: 49.9 ng/ul 1.81; ⑦+⑤D: 65.8 ng/ul 1.75; ⑧+⑤C: 73.4ng/ul 1.81; ⑧+⑤D: 72.8 ng/ul 1.81; ⑨+⑤C: 65.8 ng/ul 1.74; ⑨+⑤D: 64.1 ng/ul 1.83. | ||
| − | + | == 8.7 == | |
| − | + | === Enzyme Digestion and Go Gel Electrophoresis Test of ⑥+⑤, ⑦+⑤, ⑧+⑤, ⑨+⑤ C and D === | |
'''Time:''' 2:00 | '''Time:''' 2:00 | ||
| Line 1,467: | Line 1,467: | ||
'''Handler:''' Tang Shiqiang | '''Handler:''' Tang Shiqiang | ||
| − | + | ==== Procedure: ==== | |
# Make enzyme cutting system solution. | # Make enzyme cutting system solution. | ||
| Line 1,549: | Line 1,549: | ||
# Go gel electrophoresis. | # Go gel electrophoresis. | ||
| − | + | ==== Results: ==== | |
{{SUSTech_Image | filename=SUSTech_Shenzhen-1C4A2A62-E9CB-49BE-B41A-544A82C84E24.png | caption= | width=500px}} | {{SUSTech_Image | filename=SUSTech_Shenzhen-1C4A2A62-E9CB-49BE-B41A-544A82C84E24.png | caption= | width=500px}} | ||
| − | + | === Observation of LB medium color of ⑩+⑥+⑤, ⑩+⑦+⑤, ⑩+⑧+⑤, ⑩+⑨+⑤, 11+⑥+⑤, 11+⑦+⑤, 11+⑧+⑤, 11+⑨+⑤ === | |
'''Time:''' 7:00 | '''Time:''' 7:00 | ||
| Line 1,558: | Line 1,558: | ||
'''Handler:''' Xiao Tianyao | '''Handler:''' Xiao Tianyao | ||
| − | + | ==== Results: ==== | |
{| class=" table table-striped" | {| class=" table table-striped" | ||
| Line 1,592: | Line 1,592: | ||
‘’’Note:’’’ There are yellow colonies on plate 11+⑧+⑤ and red colonies on 11+⑨+⑤. | ‘’’Note:’’’ There are yellow colonies on plate 11+⑧+⑤ and red colonies on 11+⑨+⑤. | ||
| − | + | === Picking single colonies of ⑩+⑦+⑤, ⑩+⑧+⑤, ⑩+⑨+⑤, 11+⑥+⑤, 11+⑧+⑤, 11+⑨+⑤ (Because there are obvious correct color colonies on those plates)=== | |
'''Time:''' 9:30 | '''Time:''' 9:30 | ||
| Line 1,598: | Line 1,598: | ||
'''Handler:''' Liao Weiduo | '''Handler:''' Liao Weiduo | ||
| − | + | ==== Procedure: ==== | |
# Pick single colonies, adding 6 ml C+ LB. | # Pick single colonies, adding 6 ml C+ LB. | ||
# Incubate at 37℃ for about 12h, 220rpm. | # Incubate at 37℃ for about 12h, 220rpm. | ||
| − | + | === Ligation of ⑩ and ⑥+⑤, 11 and ⑦+⑤ === | |
'''Time:''' 11:00 | '''Time:''' 11:00 | ||
| Line 1,609: | Line 1,609: | ||
'''Handler:''' Tang Shiqiang | '''Handler:''' Tang Shiqiang | ||
| − | + | ==== Procedure: ==== | |
# Make ligation system solution. | # Make ligation system solution. | ||
| Line 1,664: | Line 1,664: | ||
# Take 5ul ligation system solution to do transformation. | # Take 5ul ligation system solution to do transformation. | ||
| − | + | === Plasmid extraction of ⑩+⑦+⑤, ⑩+⑧+⑤, ⑩+⑨+⑤, 11+⑥+⑤, 11+⑧+⑤, 11+⑨+⑤ === | |
'''Time:''' 22:00 | '''Time:''' 22:00 | ||
| Line 1,670: | Line 1,670: | ||
'''Handler:''' Liao Weiduo | '''Handler:''' Liao Weiduo | ||
| − | + | ==== Procedure: bio-tek OMEGA E.Z.N.A. Plasmid DNA Mini Kit I Spin Protocol ==== | |
| − | + | ==== Results: ==== | |
Concentration: ⑩+⑦+⑤: 63.6 ng/ul 1.71; ⑩+⑧+⑤: 100.7 ng/ul 1.54; ⑩+⑨+⑤: 83.0 ng/ul 1.59; 11+⑥+⑤: 114.8 ng/ul 1.65; 11+⑧+⑤: 77.5 ng/ul 1.78; 11+⑨+⑤: 106.3 ng/ul 1.51. | Concentration: ⑩+⑦+⑤: 63.6 ng/ul 1.71; ⑩+⑧+⑤: 100.7 ng/ul 1.54; ⑩+⑨+⑤: 83.0 ng/ul 1.59; 11+⑥+⑤: 114.8 ng/ul 1.65; 11+⑧+⑤: 77.5 ng/ul 1.78; 11+⑨+⑤: 106.3 ng/ul 1.51. | ||
| − | + | == 8.9 == | |
| − | + | === Picking single colonies of ⑩+⑥+⑤, 11+⑦+⑤ === | |
'''Time:''' 00:00 | '''Time:''' 00:00 | ||
| Line 1,684: | Line 1,684: | ||
'''Handler:''' Tang Shiqiang | '''Handler:''' Tang Shiqiang | ||
| − | + | ==== Procedure: ==== | |
# Pick single colonies, adding 6ml LB with corresponding antibiotics. | # Pick single colonies, adding 6ml LB with corresponding antibiotics. | ||
# Incubate overnight at 37℃, 220rpm. | # Incubate overnight at 37℃, 220rpm. | ||
| − | + | ==== Plasmid extraction of ⑩+⑥+⑤, 11+⑦+⑤ ==== | |
'''Time:''' 16:00 | '''Time:''' 16:00 | ||
| Line 1,695: | Line 1,695: | ||
'''Handler:''' Tang Shiqiang | '''Handler:''' Tang Shiqiang | ||
| − | + | ==== Procedure: bio-tek OMEGA E.Z.N.A. Plasmid DNA Mini Kit I Spin Protocol ==== | |
| − | + | ==== Results: ==== | |
Concentration: ⑩+⑥+⑤: 109.0 ng/ul 1.74; 11+⑦+⑤: 115.3 ng/ul 1.85. | Concentration: ⑩+⑥+⑤: 109.0 ng/ul 1.74; 11+⑦+⑤: 115.3 ng/ul 1.85. | ||
| − | + | = sfGFP notebook = | |
| − | + | == 8.2 == | |
| − | + | === Plate streaking of a plasmid with contain sfGFP and msfGFP === | |
'''Time:''' 17:30 | '''Time:''' 17:30 | ||
| Line 1,711: | Line 1,711: | ||
'''Handler:''' Tang Shiqiang | '''Handler:''' Tang Shiqiang | ||
| − | + | == 8.3 == | |
| − | + | === Picking single colonies of sfGFP and msfGFP === | |
'''Time:''' 9:00 | '''Time:''' 9:00 | ||
| Line 1,719: | Line 1,719: | ||
'''Handler:''' Tang Shiqiang | '''Handler:''' Tang Shiqiang | ||
| − | + | ==== Procedure: ==== | |
# Pick single colonies, adding 6ml LB with corresponding antibiotics. | # Pick single colonies, adding 6ml LB with corresponding antibiotics. | ||
# Incubate for about 12h at 37℃, 220rpm. | # Incubate for about 12h at 37℃, 220rpm. | ||
| − | + | === Plasmid extraction of sfGFP and msfGFP === | |
'''Time:''' 21:30 | '''Time:''' 21:30 | ||
| Line 1,730: | Line 1,730: | ||
'''Handler:''' Lu Shixin | '''Handler:''' Lu Shixin | ||
| − | + | ==== Procedure:bio-tek OMEGA E.Z.N.A. Plasmid DNA Mini Kit I Spin Protocol ==== | |
| − | + | ==== Result: ==== | |
Concentration: sfGFP: 70.0 ng/ul 1.77; msfGFP: 460.8 ng/ul. | Concentration: sfGFP: 70.0 ng/ul 1.77; msfGFP: 460.8 ng/ul. | ||
| − | + | == 8.16 == | |
| − | + | === PCR of sfGFP and msfGFP === | |
'''Time:''' 16:00 | '''Time:''' 16:00 | ||
| Line 1,744: | Line 1,744: | ||
'''Handler:''' Tang Shiqiang | '''Handler:''' Tang Shiqiang | ||
| − | + | ==== Procedure: ==== | |
{|class="table table-striped" | {|class="table table-striped" | ||
| Line 1,792: | Line 1,792: | ||
|} | |} | ||
| − | + | === Go Gel Electrophoresis and Gel Extraction of sfGFP and msfGFP PCR product === | |
'''Time:''' 19:00 | '''Time:''' 19:00 | ||
| Line 1,798: | Line 1,798: | ||
'''Handler:''' Tang Shiqiang | '''Handler:''' Tang Shiqiang | ||
| − | + | ==== Procedure: bio-tek OMEGA E.Z.N.A. Gel Extraction Kit – Spin Protocol ==== | |
| − | + | ==== Result: ==== | |
Concentration: sfGFP: 43.2 ng/ul 1.76; msfGFP: 62.0 ng/ul 1.79. | Concentration: sfGFP: 43.2 ng/ul 1.76; msfGFP: 62.0 ng/ul 1.79. | ||
| − | + | === Enzyme Digestion for Gel Extraction of sfGFP and msfGFP === | |
'''Time:''' 19:30 | '''Time:''' 19:30 | ||
| Line 1,810: | Line 1,810: | ||
'''Handler:''' Tang Shiqiang | '''Handler:''' Tang Shiqiang | ||
| − | + | ==== Procedure: ==== | |
# Make enzyme cutting system solution. | # Make enzyme cutting system solution. | ||
| Line 1,846: | Line 1,846: | ||
# Incubate at 37℃ for 3h. | # Incubate at 37℃ for 3h. | ||
| − | + | === Enzyme Digestion for Gel Extraction of ⑤ === | |
'''Time:''' 17:00 | '''Time:''' 17:00 | ||
| Line 1,852: | Line 1,852: | ||
'''Handler:''' Tang Shiqiang | '''Handler:''' Tang Shiqiang | ||
| − | + | ==== Procedure: ==== | |
# Make enzyme cutting system solution. | # Make enzyme cutting system solution. | ||
| Line 1,882: | Line 1,882: | ||
# Incubate at 37℃ for 3h | # Incubate at 37℃ for 3h | ||
| − | + | === Go Gel Electrophoresis and Gel Extraction of sfGFP, msfGFP, and ⑤ === | |
'''Time:''' 20:00 | '''Time:''' 20:00 | ||
| Line 1,888: | Line 1,888: | ||
'''Handler:''' Tang Shiqiang | '''Handler:''' Tang Shiqiang | ||
| − | + | ==== Procedure: bio-tek OMEGA E.Z.N.A. Gel Extraction Kit – Spin Protocol ==== | |
| − | + | ==== Results:==== | |
[[File:media/image1.png|145x256px]] | [[File:media/image1.png|145x256px]] | ||
| Line 1,896: | Line 1,896: | ||
Gel extraction concentration: ⑤: 61.4 ng/ul 1.58; sfGFP: 16.9 ng/ul 1.62; msfGFP: 16.5 ng/ul 1.59. | Gel extraction concentration: ⑤: 61.4 ng/ul 1.58; sfGFP: 16.9 ng/ul 1.62; msfGFP: 16.5 ng/ul 1.59. | ||
| − | + | == 8.17== | |
| − | + | === Ligation of sfGFP+⑤, msfGFP+⑤, sfGFP+pSB1C3, msfGFP+pSB1C3 === | |
'''Time:''' 0:00 | '''Time:''' 0:00 | ||
| Line 1,904: | Line 1,904: | ||
'''Handler:''' Tang Shiqiang | '''Handler:''' Tang Shiqiang | ||
| − | + | ==== Procedure:==== | |
# Make ligation system solution. | # Make ligation system solution. | ||
| Line 1,942: | Line 1,942: | ||
# Take 10.5ul ligation system solution to do transformation. | # Take 10.5ul ligation system solution to do transformation. | ||
| − | + | === Picking single colonies of sfGFP and msfGFP === | |
'''Time:''' 18:30 | '''Time:''' 18:30 | ||
| Line 1,948: | Line 1,948: | ||
'''Handler:''' Tang Shiqiang | '''Handler:''' Tang Shiqiang | ||
| − | + | ==== Procedure:==== | |
# Pick single colonies, adding 6ml LB with corresponding antibiotics. | # Pick single colonies, adding 6ml LB with corresponding antibiotics. | ||
# Incubate overnight at 37℃, 220rpm. | # Incubate overnight at 37℃, 220rpm. | ||
| − | + | == 8.18 == | |
| − | + | === Plasmid extraction of sfGFP+⑤, msfGFP+⑤, sfGFP+pSB1C3, msfGFP+pSB1C3 === | |
'''Time:''' 13:00 | '''Time:''' 13:00 | ||
| Line 1,961: | Line 1,961: | ||
'''Handler:''' Tang Shiqiang | '''Handler:''' Tang Shiqiang | ||
| − | + | ==== Procedure: bio-tek OMEGA E.Z.N.A. Plasmid DNA Mini Kit I Spin Protocol ==== | |
| − | + | ==== Result:==== | |
Concentration: sfGFP+⑤: 103.6 ng/ul 1.96; msfGFP+⑤: 127.1 ng/ul 1.80; sfGFP+pSB1C3①: 156.1 ng/ul 1.91; sfGFP+pSB1C3②: 179.2 ng/ul 1.91; msfGFP+pSB1C3①: 115.5 ng/ul 1.93; msfGFP+pSB1C3②: 159.5 ng/ul 1.92. | Concentration: sfGFP+⑤: 103.6 ng/ul 1.96; msfGFP+⑤: 127.1 ng/ul 1.80; sfGFP+pSB1C3①: 156.1 ng/ul 1.91; sfGFP+pSB1C3②: 179.2 ng/ul 1.91; msfGFP+pSB1C3①: 115.5 ng/ul 1.93; msfGFP+pSB1C3②: 159.5 ng/ul 1.92. | ||
| − | + | == 8.18 == | |
| − | + | === Enzyme Digestion and Go Gel Electrophoresis Test of sfGFP+⑤, msfGFP+⑤, sfGFP+pSB1C3, msfGFP+pSB1C3 === | |
'''Time:''' 0:00 | '''Time:''' 0:00 | ||
| Line 1,975: | Line 1,975: | ||
'''Handler:''' Tang Shiqiang | '''Handler:''' Tang Shiqiang | ||
| − | + | ==== Procedure: ==== | |
# Make enzyme cutting system solution. | # Make enzyme cutting system solution. | ||
| Line 2,145: | Line 2,145: | ||
# Go gel electrophoresis. | # Go gel electrophoresis. | ||
| − | + | ==== Results: ==== | |
[[File:media/image2.png|553x273px]] | [[File:media/image2.png|553x273px]] | ||
| − | + | == 9.21 == | |
| − | + | === PCR of msfGFP Coding sequence basic part === | |
'''Time:''' 16:00 | '''Time:''' 16:00 | ||
| Line 2,157: | Line 2,157: | ||
'''Handler:''' Tang Shiqiang | '''Handler:''' Tang Shiqiang | ||
| − | + | ==== Procedure: ==== | |
{|class="table table-striped" | {|class="table table-striped" | ||
| Line 2,205: | Line 2,205: | ||
|} | |} | ||
| − | + | === Go Gel Electrophoresis and Gel Extraction of msfGFP CDS === | |
'''Time:''' 20:00 | '''Time:''' 20:00 | ||
| Line 2,211: | Line 2,211: | ||
'''Handler:''' Tang Shiqiang | '''Handler:''' Tang Shiqiang | ||
| − | + | ==== Procedure: bio-tek OMEGA E.Z.N.A. Gel Extraction Kit – Spin Protocol ==== | |
| − | + | ==== Result: ==== | |
Concentration: msfGFP: 96.1 ng/ul 1.83. | Concentration: msfGFP: 96.1 ng/ul 1.83. | ||
| − | + | === Enzyme Digestion for Gel Extraction of msfGFP CDS === | |
'''Time:''' 20:30 | '''Time:''' 20:30 | ||
| Line 2,223: | Line 2,223: | ||
'''Handler:''' Tang Shiqiang | '''Handler:''' Tang Shiqiang | ||
| − | + | ==== Procedure: ==== | |
# Make enzyme cutting system solution. | # Make enzyme cutting system solution. | ||
| Line 2,253: | Line 2,253: | ||
# Incubate overnight at 37℃. | # Incubate overnight at 37℃. | ||
| − | + | == 9.22 == | |
| − | + | === Transformation of msfGFP CDS === | |
'''Time:''' 11:00 | '''Time:''' 11:00 | ||
| Line 2,261: | Line 2,261: | ||
'''Handler:''' Tang Shiqiang | '''Handler:''' Tang Shiqiang | ||
| − | + | ==== Procedure: IGEM Protocols ==== | |
| − | + | === Picking single colonies of sfGFP CDS === | |
'''Time:''' 23:00 | '''Time:''' 23:00 | ||
| Line 2,269: | Line 2,269: | ||
'''Handler:''' Tang Shiqiang | '''Handler:''' Tang Shiqiang | ||
| − | + | ==== Procedure: ==== | |
# Pick single colonies, adding 5ml C+ LB. | # Pick single colonies, adding 5ml C+ LB. | ||
# Incubate overnight at 37℃, 220rpm. | # Incubate overnight at 37℃, 220rpm. | ||
| − | + | == 9.23 == | |
| − | + | === Plasmid extraction of msfGFP CDS === | |
'''Time:''' 17:00 | '''Time:''' 17:00 | ||
| Line 2,282: | Line 2,282: | ||
'''Handler:''' Tang Shiqiang | '''Handler:''' Tang Shiqiang | ||
| − | + | ==== Procedure: bio-tek OMEGA E.Z.N.A. Plasmid DNA Mini Kit I Spin Protocol ==== | |
| − | + | ==== Result: ==== | |
Concentration: msfGFP CDS①: 141.0 ng/ul 1.88; msfGFP CDS②: 141.0 ng/ul 1.90. | Concentration: msfGFP CDS①: 141.0 ng/ul 1.88; msfGFP CDS②: 141.0 ng/ul 1.90. | ||
| − | + | == 9.24 == | |
| − | + | === Enzyme Digestion and Go Gel Electrophoresis Test of msfGFP CDS① and msfGFP CDS② === | |
'''Time:''' 11:40 | '''Time:''' 11:40 | ||
| Line 2,296: | Line 2,296: | ||
'''Handler:''' Tang Shiqiang | '''Handler:''' Tang Shiqiang | ||
| − | + | ==== Procedure: ==== | |
# Make enzyme cutting system solution. | # Make enzyme cutting system solution. | ||
| Line 2,332: | Line 2,332: | ||
# Go gel electrophoresis. | # Go gel electrophoresis. | ||
| − | + | ==== Results: ==== | |
[[File:media/image3.png|389x357px]] | [[File:media/image3.png|389x357px]] | ||
| + | |||
{{:Team:SUSTech_Shenzhen/main-content-end}} | {{:Team:SUSTech_Shenzhen/main-content-end}} | ||
{{:Team:SUSTech_Shenzhen/wiki-footer}} | {{:Team:SUSTech_Shenzhen/wiki-footer}} | ||
{{:Team:SUSTech_Shenzhen/themeJs}} | {{:Team:SUSTech_Shenzhen/themeJs}} | ||
Revision as of 01:13, 13 October 2016

Biobricks
Notebook
Contents
- 1 8 Chromoprotein
- 1.1 Plasmid Construction
- 1.2 7.22
- 1.3 7.23
- 1.4 7.24
- 1.5 7.25
- 1.6 7.26
- 1.7 7.27
- 1.8 7.28
- 1.9 7.29
- 1.10 7.30
- 1.10.1 Picking single colonies of ⑧, ⑩, 11
- 1.10.2 Go Gel Electrophoresis and Gel Extraction of ⑤,⑥,⑦,⑧,⑨
- 1.10.3 Enzyme Digestion for Gel Extraction of ⑤,⑥,⑦,⑨ (Redo)
- 1.10.4 Plasmid extraction of ⑧,⑩,11
- 1.10.5 Enzyme Digestion for Gel Extraction of ⑧,⑩,11
- 1.10.6 Go Gel Electrophoresis and Gel Extraction of ⑤,⑥,⑦,⑨ (Redo)
- 1.11 7.31
- 1.12 8.1
- 1.13 8.2
- 1.14 8.3
- 1.15 8.4
- 1.16 8.5
- 1.17 8.6
- 1.18 8.7
- 1.18.1 Enzyme Digestion and Go Gel Electrophoresis Test of ⑥+⑤, ⑦+⑤, ⑧+⑤, ⑨+⑤ C and D
- 1.18.2 Observation of LB medium color of ⑩+⑥+⑤, ⑩+⑦+⑤, ⑩+⑧+⑤, ⑩+⑨+⑤, 11+⑥+⑤, 11+⑦+⑤, 11+⑧+⑤, 11+⑨+⑤
- 1.18.3 Picking single colonies of ⑩+⑦+⑤, ⑩+⑧+⑤, ⑩+⑨+⑤, 11+⑥+⑤, 11+⑧+⑤, 11+⑨+⑤ (Because there are obvious correct color colonies on those plates)
- 1.18.4 Ligation of ⑩ and ⑥+⑤, 11 and ⑦+⑤
- 1.18.5 Plasmid extraction of ⑩+⑦+⑤, ⑩+⑧+⑤, ⑩+⑨+⑤, 11+⑥+⑤, 11+⑧+⑤, 11+⑨+⑤
- 1.19 8.9
- 2 sfGFP notebook
- 2.1 8.2
- 2.2 8.3
- 2.3 8.16
- 2.4 8.17
- 2.5 8.18
- 2.6 8.18
- 2.7 9.21
- 2.8 9.22
- 2.9 9.23
- 2.10 9.24
8 Chromoprotein
Plasmid Construction
ALL ABBREVIATIONS USED:
| Name | Number | Index | Position | Backbone | Length |
|---|---|---|---|---|---|
| Strong promoter |
|
BBa_J23100 | 4-17-D | BBa_J61002 | 35 |
| Weak promoter |
|
BBa_J23106 | 4-17-P | BBa_J61002 | 35 |
| Strong RBS | ③ | BBa_B0034 | 4-1-N | pSB1A2 | 12 |
| Weak RBS | ④ | BBa_B0031 | 2-2-H | pSB1C3 | 14 |
| Terminator | ⑤ | BBa_B0015 | 3-3-F | pSB1C3 | 129 |
| eforRed | ⑥ | BBa_K592012 | 6-15-I | pSB1C3 | 681 |
| gfasPurple | ⑦ | BBa_K1033919 | 6-9-K | pSB1C3 | 669 |
| FwYellow | ⑧ | BBa_K1033910 | 4-6-K | pSB1C3 | 714 |
| SpisPink | ⑨ | BBa_K1033932 | 6-11-k | pSB1C3 | 678 |
| ①+③ | ⑩ | BBa_K880005 | 2-3-F | pSB1C3 | 55 |
| Medium promoter+④ | 11 | BBa_K608007 | 1-5-G | pSB1C3 | 57 |
7.22
Transformation of ①, ③, ⑤
Time: 20:30
Handler: Liao Weiduo
Procedure: IGEM Protocols
1. Thaw competent cells on ice.
2. Punch a hole into the corresponding hole in the hole in 2016 kit plate by a 10 ul pipette tip. Adding 10ul ddH2O to the hole and pipette up and down for several times. Let sit for several minutes to make sure the dyed DNA is fully resuspended.
3. Pipette 50ul of competent cells into 1.5ml tube.
4. Pipette 1ul of resuspended DNA into 1.5ml tube.
5. Pipette 1ul of ddH2O as a control into 1.5ml tube.
6. Close 1.5ml tubes, incubate on ice for 30 min.
7. Heat shock tubes at 42℃ for 1 min.
8. Incubate on ice for 5 min.
9. Pipette 200ul SOC media to each transformation.
10. Incubate at 37℃ for 2 hours, 220rpm.
11. Pipette each transformation on petri plates for a 100ul, and spread with sterilized spreader immediately.
12. Incubate transformations overnight at 37℃.
7.23
Picking single colonies of ①, ③, ⑤
Time: 16:00
Handler: Liao Weiduo
Procedure:
- Pick single colonies, adding 6ml LB with corresponding antibiotics, each plate we pick 2 single colonies, A and B.
- Incubate overnight at 37℃, 220rpm.
7.24
Plasmid extraction of ①, ③, ⑤ A and B
Time: 9:00
Handler: Liao Weiduo
Procedure: bio-tek OMEGA E.Z.N.A. Plasmid DNA Mini Kit I Spin Protocol
- Add 2ml LB medium to a 2ml microcentrifuge tube.
- Centrifuge at 10,000 g for 1 minute at roonm temperature.
- Discard the supernatant.
- Repeat Step1-3 until 4ml sample has been transferred to the 2ml tube.
- Add 250ul Solution I/RNase A. Vortex to mix thoroughly.
- Add 250ul Solution II. Invert and gently rotate the tube several times to obtain a clear lysate. A 2-3 minute incubation may be necessary.
- Add 350ul Solution III. Immediately invert several times until a flocculent white precipitate forms.
- Centrifuge at maximum speed (>13,000 g) for 10 minutes.
- Insert a HiBind DNA Mini Column into a 2ml Collection Tube.
- Transfer the cleared supernatant (~830ul)from Step 8 by CAREFULLY aspirating it into the HiBind DNA Mini Column.
- Centrifuge at maximum speed for 1 minute.
- Discard the filtrate and reuse the collection tube.
- Add 500ul HBC Buffer. (HBC Buffer must be diluted with isopropanol before use)
- Centrifuge at maximum speed for 1 min.
- Discard the filtrate and reuse collection tube.
- Add 700ul DNA Wash Buffer. (DNA Wash Buffer must be diluted with 100% ethanol prior to use.)
- Centrifuge at maximum speed for 1 min.
- Discard the filtrate and reuse the collection tube.
- Repeat Steps 16-18.
- Centrifuge the empty HiBind DNA Mini Column for 2 minutes at maximum speed to dry the column matrix.
- Transfer the HiBind DNA Mini Column to a clean 1.5ml microcentrifuge tube.
- Add 80ul ddH2O.
- Let sit at room temperature for 2 min.
- Centrifuge at maximum speed for 1 min.
Results:
‘’’Concentration:’’’ ①A: 245.9 ng/ul 1.84; ①B: 340.5 ng/ul 1.84; ③A: 116.2 ng/ul 1.65; ③B: 102.1 ng/ul 1.77; ⑤A: 151.4 ng/ul 1.80; ⑤B: 68.0 ng/ul 1.84;
7.25
Transformation of ②,④,⑥,⑦,⑧,⑨
Time: 0:00
Handler: Tang Shiqiang
Procedure: IGEM Protocols
Picking single colonies of ②,④,⑥,⑦,⑧,⑨
Time: 20:00
Handler: Tang Shiqiang
Procedure:
- Pick single colonies, adding 6ml LB with corresponding antibiotics, each plate we pick 2 single colonies, α and β.
- Incubate overnight at 37℃, 220rpm.
7.26
Plasmid extraction of ②,④,⑥,⑦,⑧,⑨ group α
Time: 9:40
Handler: Lu Shixin
Procedure: bio-tek OMEGA E.Z.N.A. Plasmid DNA Mini Kit I Spin Protocol
Results:
Concentration: ②α: 369.8 ng/ul; ④α: 142.4 ng/ul; ⑥α: 116.5 ng/ul; ⑦α: 92 ng/ul; ⑧α: 157.4 ng/ul; ⑨α: 168.3 ng/ul.
‘’’ Note: ’’’ ⑧ has a red colony.
Plasmid extraction of ②,④,⑥,⑦,⑧,⑨ group β
Time: 14:00
Handler: Tang Shiqiang
Procedure: bio-tek OMEGA E.Z.N.A. Plasmid DNA Mini Kit I Spin Protocol
Results:
Concentration: ②β: 496.9 ng/ul 1.86; ④β: 256.4 ng/ul 1.85; ⑥β: 220.3 ng/ul 1.85; ⑦β: 225.7 ng/ul 1.85; ⑧β: 304.6 ng/ul 1.85; ⑨β: 352.2 ng/ul 1.84.
‘’’ Note: ‘’’ ⑧ has a red colony.
7.27
Competent Cell Test
Time: 16:00
Handler: Lu Shixin
Procedure: IGEM Protocols
- Spin down the DNA tubes form the Competent Cell Test Kit to collect all of the DNA into the bottom of each tube prior to use.
- Thaw competent cells on ice.
- Pipet 50ul of competent cells into each tube.
- Pipet 1ul of DNA into each microcentrifuge tube.
- Incubate on ice for 30 min.
- Heat shock at 42℃ for 1 min.
- Incubate on ice for 5 min.
- Add 200ul SOC media, incubate at 37℃ for 2h.
- Pipet 20ul from each tube onto the appropriate plate, and spread it.
- Incubate at 37℃ overnight, approximately 16 h.
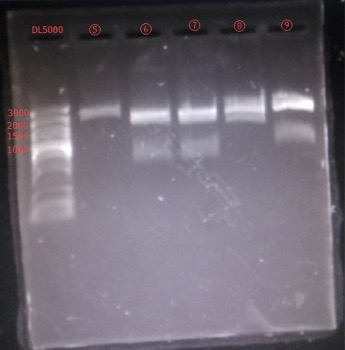
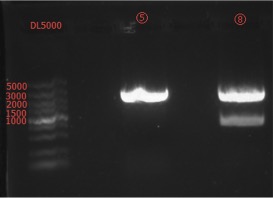
Enzyme Digestion and Go Gel Electrophoresis Test of ②,④,⑥,⑦,⑧,⑨
Time: 20:00
Handler: Xiao Tianyao
Procedure:
1. Make enzyme cutting system solution.
| ②α | ④α | ⑥α | ⑦α | ⑧α | ⑨α | |
|---|---|---|---|---|---|---|
| DNA(400ng) | 1ul | 2ul | 2ul | 2ul | 1.2ul | 1.2ul |
| Cutsmart buffer | 2ul | 2ul | 2ul | 2ul | 2ul | 2ul |
| EcoRI-HF | 0.2ul | 0.2ul | 0.2ul | 0.2ul | 0.2ul | 0.2ul |
| PstI-HF | 0.2ul | 0.2ul | 0.2ul | 0.2ul | 0.2ul | 0.2ul |
| ddH2O | 16.6ul | 15.6ul | 15.6ul | 15.6ul | 16.4ul | 16.4ul |
| Total | 20ul | 20ul | 20ul | 20ul | 20ul | 20ul |
| ②β | ④β | ⑥β | ⑦β | ⑧β | ⑨β | |
|---|---|---|---|---|---|---|
| DNA(400ng) | 1ul | 2ul | 2ul | 2ul | 1.2ul | 1.2ul |
| Cutsmart buffer | 2ul | 2ul | 2ul | 2ul | 2ul | 2ul |
| EcoRI-HF | 0.2ul | 0.2ul | 0.2ul | 0.2ul | 0.2ul | 0.2ul |
| PstI-HF | 0.2ul | 0.2ul | 0.2ul | 0.2ul | 0.2ul | 0.2ul |
| ddH2O | 16.6ul | 15.6ul | 15.6ul | 15.6ul | 16.4ul | 16.4ul |
| Total | 20ul | 20ul | 20ul | 20ul | 20ul | 20ul |
Incubate at 37℃ for 30 min.
Make 1% agarose gel 30ml.
Add 6x loading dye to the enzyme cutting system solution.
Go gel electrophoresis.
Results:
figures show below:
7.28
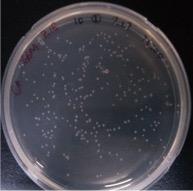
Competent Cell Test Colonies Counting
Time: 13:00
Handler: Lu Shixin
Result:
And all the colonies are red under UV light.
7.29
Transformation of ⑧,⑩,11
Time: 16:00
Handler: Lu Shixin, Tang Shiqiang
Procedure: IGEM Transformation Protocol
Enzyme Digestion for Gel Extraction of ⑤,⑥,⑦,⑧,⑨
Time: 20:00
Handler: LuShixin, Tang Shiqiang
Procedure:
- Make enzyme cutting system solution.
| ⑥ | ⑦ | ⑧ | ⑨ | |
|---|---|---|---|---|
| DNA(500ng) | 2.5ul | 2.5ul | 2.0ul | 1.5ul |
| Cutsmart buffer | 2.5ul | 2.5ul | 2.5ul | 2.5ul |
| EcoRI-HF | 0.5ul | 0.5ul | 0.5ul | 0.5ul |
| SpeI-HF | 0.5ul | 0.5ul | 0.5ul | 0.5ul |
| ddH2O | 19ul | 19ul | 19.5ul | 20ul |
| Total | 25ul | 25ul | 25ul | 25ul |
| ⑤ | |
|---|---|
| DNA (500ng) | 8ul |
| Cutsmart buffer | 2.5ul |
| EcoRI-HF | 0.5ul |
| XbaI | 0.5ul |
| ddH2O | 16.5ul |
| Total | 25ul |
- Incubate at 37℃ overnight. (Start from 21:20)
7.30
Picking single colonies of ⑧, ⑩, 11
Time: 9:00
Handler: Lu Shixin
Procedure:
- Pick single colonies, adding 1ml LB with corresponding antibiotics in 1.5ml EP tube, each plate we pick 2 single colonies, α and β.
- Incubate for 2h at 37℃.
- Transfer 1ml LB from 1.5ml EP tube to 14ml bacteria shaking tube, and add 5ml corresponding antibiotics LB.
- Incubate at 37℃ for 10h, 220rpm.
Go Gel Electrophoresis and Gel Extraction of ⑤,⑥,⑦,⑧,⑨
Time: 9:00
Handler: Lu Shixin
Procedure: bio-tek OMEGA E.Z.N.A. Gel Extraction Kit – Spin Protocol
- Make 1% agarose gel 30ml.
- Add 6x loading dye to the enzyme cutting system solution.
- Go gel electrophoresis.
- Excise the DNA fragment of interest.
- Determine the volume of the gel slice by weighing it in a clean 1.5ml EP tube.
- Add 1 volume Binding Buffer (XP2).
- Incubate at 50-60℃ for 7 min or until the gel has completely melted. Vortex or shake the tube every 2-3 min.
- Insert a HiBind DNA Mini Column in a 2ml Collection Tube.
- Add no more than 700ul DNA/agarose solution from Step 7 to the HiBind DNA Mini Column.
- Centrifuge at 10,000 g for 1 min at room temperature.
- Discard the filtrate and reuse collection tube.
- Repeat Steps 9-11 until all of the sample has been transferred to the column.
- Add 300ul Binding Buffer(XP2).
- Centrifuge at maximum speed (>13,000 g) for 1 min at room temperature.
- Discard the filtrate and reuse collection tube.
- Add 700ul SPW Wash Buffer. (SPW Wash Buffer must be diluted with 100% ethanol prior to use.)
- Centrifuge at maximum speed for 1 min at room temperature.
- Discard the filtrate and reuse collection tube.
- Repeat Steps 16-18.
- Centrifuge the empty HiBind DNA Mini Column for 2 min at maximum speed to dry the column matrix.
- Transfer the HiBind DNA Mini Column to a clean 1.5ml EP tube.
- Add 15-30ul ddH2O.
- Let sit at room temperature for 2 min.
- Centrifuge at maximum speed for 1 min.
Results:
All the concentration of gel extraction are below 10 ng/ul. So we decide to repeat enzyme digestion with more DNA.
Enzyme Digestion for Gel Extraction of ⑤,⑥,⑦,⑨ (Redo)
Time: 18:00
Handler: Lu Shixin
Procedure:
- Make enzyme cutting system solution.
| ⑥ | ⑦ | ⑨ | |
|---|---|---|---|
| DNA(>4ug) | 21.5ul | 21.5ul | 21.5ul |
| Cutsmart buffer | 2.5ul | 2.5ul | 2.5ul |
| EcoRI-HF | 0.5ul | 0.5ul | 0.5ul |
| SpeI-HF | 0.5ul | 0.5ul | 0.5ul |
| ddH2O | 0ul | 0ul | 0ul |
| Total | 25ul | 25ul | 25ul |
| ⑤ | |
|---|---|
| DNA (2ug) | 21.5ul |
| Cutsmart buffer | 2.5ul |
| EcoRI-HF | 0.5ul |
| XbaI | 0.5ul |
| ddH2O | 0ul |
| Total | 25ul |
- Incubate at 37℃ for 3h. (Start from 18:40)
Plasmid extraction of ⑧,⑩,11
Time: 21:00
Handler: Lu Shixin
Procedure: bio-tek OMEGA E.Z.N.A. Plasmid DNA Mini Kit I Spin Protocol
Results:
The length of them are correct.
Concentration: ⑧: 130.2 ng/ul; ⑩: 147.0 ng/ul; 11: 93.3 ng/ul.
Enzyme Digestion for Gel Extraction of ⑧,⑩,11
Time: 22:30
Handler: Lu Shixin
Procedure:
- Make enzyme cutting system solution.
| ⑩ | 11 | |
|---|---|---|
| DNA(>1.5ug) | 11.5ul | 21.5ul |
| Cutsmart buffer | 2.5ul | 2.5ul |
| SpeI-HF | 0.5ul | 0.5ul |
| PstI-HF | 0.5ul | 0.5ul |
| ddH2O | 10ul | 0ul |
| Total | 25ul | 25ul |
| ⑧ | |
|---|---|
| DNA (0.5ug) | 4ul |
| Cutsmart buffer | 2.5ul |
| EcoRI-HF | 0.5ul |
| XbaI | 0.5ul |
| ddH2O | 17.5ul |
| Total | 25ul |
- Incubate at 37℃ overnight. (Start from 22:40)
Go Gel Electrophoresis and Gel Extraction of ⑤,⑥,⑦,⑨ (Redo)
Time: 22:00
Handler: Tang Shiqiang
Procedure: bio-tek OMEGA E.Z.N.A. Gel Extraction Kit – Spin Protocol
Results:
Gel extraction concentration: ⑤: 15.3 ng/ul; ⑥: 36.3 ng/ul; ⑦: 36.8 ng/ul; ⑨: 26.9 ng/ul.
7.31
Ligation of ⑥+⑤, ⑦+⑤, ⑨+⑤
Time: 0:00
Handler: Tang Shiqiang
Procedure:
- Make ligation system solution.
| ⑥+⑤ | ⑦+⑤ | ⑨+⑤ | |
|---|---|---|---|
| Vector (50ng) | 3.3ul | 3.3ul | 3.3ul |
| Insert | 1.4ul | 1.4ul | 1.9ul |
| ddH2O | 5.3ul | 5.3ul | 4.8ul |
| 2x Quick ligase buffer | 10ul | 10ul | 10ul |
| Quick ligase | 1ul | 1ul | 1ul |
| Total | 21ul | 21ul | 21ul |
- Incubate at room temperature for 5 min.
- Take 2.5ul ligation system solution to do transformation.
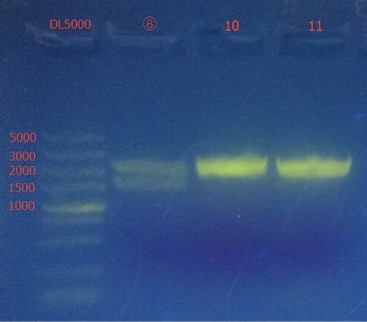
Go Gel Electrophoresis and Gel Extraction of ⑧, ⑩, 11
Time: 15:30
Handler: Lu Shixin
Procedure: bio-tek OMEGA E.Z.N.A. Gel Extraction Kit – Spin Protocol
Results:
Gel extraction concentration: ⑩: 67.3 ng/ul 1.65; 11: 66.4 ng/ul 1.69.
‘’‘Note:’‘’ We found that we take a wrong ⑧ before.
Transformation of ⑧
Time: 16:30
Handler: Tang Shiqiang
Procedure: IGEM Transformation Protocol
Picking single colonies of ⑥+⑤, ⑦+⑤, ⑨+⑤
Time: 19:30
Handler: Tang Shiqiang
Procedure:
- Pick single colonies, adding 6ml LB with corresponding antibiotics, each plate we pick 2 single colonies, A and B.
- Incubate overnight at 37℃, 220rpm.
8.1
Picking single colonies of ⑧
Time: 9:30
Handler: Tang Shiqiang
Procedure:
- Pick single colonies, adding 6ml LB with corresponding antibiotics, each plate we pick 2 single colonies, A and B.
- Incubate at 37℃ for about 14h, 220rpm.
Plasmid extraction of ⑥+⑤, ⑦+⑤, ⑨+⑤ A and B
Time: 9:30
Handler: Tang Shiqiang
Procedure: bio-tek OMEGA E.Z.N.A. Plasmid DNA Mini Kit I Spin Protocol
Results:
Concentration: ⑥+⑤A: 126.3 ng/ul 1.86; ⑥+⑤B: 124.9 ng/ul 1.82; ⑦+⑤A: 116.5 ng/ul 1.87; ⑦+⑤B: 112.0 ng/ul 1.86; ⑨+⑤A: 120.4 ng/ul 1.87; ⑨+⑤B: 149.0 ng/ul 1.87;
Enzyme Digestion and Go Gel Electrophoresis Test of ⑥+⑤, ⑦+⑤, ⑨+⑤ A and B
Time: 12:00
Handler: Tang Shiqiang
Procedure:
- Make enzyme cutting system solution.
| ⑥+⑤A | ⑦+⑤A | ⑨+⑤A | ⑥+⑤B | ⑦+⑤B | ⑨+⑤B | |
|---|---|---|---|---|---|---|
| DNA(200ng) | 2ul | 2ul | 2ul | 2ul | 2ul | 2ul |
| Cutsmart buffer | 2ul | 2ul | 2ul | 2ul | 2ul | 2ul |
| EcoRI-HF | 0.2ul | 0.2ul | 0.2ul | 0.2ul | 0.2ul | 0.2ul |
| PstI-HF | 0.2ul | 0.2ul | 0.2ul | 0.2ul | 0.2ul | 0.2ul |
| ddH2O | 15.6ul | 15.6ul | 15.6ul | 15.6ul | 15.6ul | 15.6ul |
| Total | 20ul | 20ul | 20ul | 20ul | 20ul | 20ul |
- Incubate at 37℃ for 30 min.
- Make 1% agarose gel 30ml.
- Add 6x loading dye to the enzyme cutting system solution.
- Go gel electrophoresis.
Results:
‘’‘Comments:’‘’ The length of them are incorrect. Their length are larger than 1000bp while should be smaller than 1000bp.
‘’‘Note:’‘’ Finally we found that we used a tube of wrong ⑤.
Put ⑧ A and B at 4℃
Time: 20:00
Handler: Tang Shiqiang
8.2
Plasmid extraction of ⑧ A and B
Time: 14:00
Handler: Tang Shiqiang
Procedure: bio-tek OMEGA E.Z.N.A. Plasmid DNA Mini Kit I Spin Protocol
Results:
Concentration: ⑧A: 116.5 ng/ul 1.82; ⑧B: 73.2 ng/ul 1.81.
Enzyme Digestion for Gel Extraction of ⑤, ⑧
Time: 22:00
Handler: Lu Shixin, Tang Shiqiang
Procedure:
- Make enzyme cutting system solution.
| ⑤3-3-F 1 | |
|---|---|
| DNA(>3ug) | 21.5ul |
| Cutsmart buffer | 2.5ul |
| EcoRI-HF | 0.5ul |
| XbaI | 0.5ul |
| ddH2O | 0ul |
| Total | 25ul |
| ⑧A | |
|---|---|
| DNA (>2ug) | 21.5ul |
| Cutsmart buffer | 2.5ul |
| EcoRI-HF | 0.5ul |
| SpeI-HF | 0.5ul |
| ddH2O | 0ul |
| Total | 25ul |
- Incubate at 37℃ overnight. (Start from 22:25)
8.3
Go Gel Electrophoresis and Gel Extraction of ⑤, ⑧
Time: 10:00
Handler: Lu Shixin
Procedure: bio-tek OMEGA E.Z.N.A. Gel Extraction Kit – Spin Protocol
Results:
Concentration: ⑤: 51.4 ng/ul 1.75; ⑧: 23.2 ng/ul 1.71.
Ligation of ⑥+⑤, ⑦+⑤, ⑧+⑤, ⑨+⑤
Time: 16:30
Handler: Lu Shixin
Procedure:
- Make ligation system solution.
| ⑥+⑤ | ⑦+⑤ | ⑧+⑤ | ⑨+⑤ | |
|---|---|---|---|---|
| Vector (50ng) | 1ul | 1ul | 1ul | 1ul |
| Insert | 1.5ul | 1.5ul | 2.5ul | 2ul |
| ddH2O | 7.5ul | 7.5ul | 6.5ul | 7ul |
| 2x Quick ligase buffer | 10ul | 10ul | 10ul | 10ul |
| Quick ligase | 1ul | 1ul | 1ul | 1ul |
| Total | 21ul | 21ul | 21ul | 21ul |
- Incubate at room temperature for 10 min.
- Take 2ul ligation system solution to do transformation.
8.4
Picking single colonies of ⑥+⑤, ⑦+⑤, ⑧+⑤, ⑨+⑤
Time: 9:30
Handler: Lu Shixin
Procedure:
- Pick single colonies, adding 6ml LB with corresponding antibiotics, each plate we pick 2 single colonies, A and B.
- Incubate at 37℃ for about 12h, 220rpm.
Plasmid extraction of ⑥+⑤, ⑦+⑤, ⑧+⑤, ⑨+⑤ A and B
Time: 21:30
Handler: Tang Shiqiang
Procedure: bio-tek OMEGA E.Z.N.A. Plasmid DNA Mini Kit I Spin Protocol
Results:
Concentration: ⑥+⑤A: 54.4 ng/ul 2.18; ⑥+⑤B: 29.2 ng/ul 3.15; ⑦+⑤A: 55.4 ng/ul 2.42; ⑦+⑤B: 58.8 ng/ul 2.42; ⑧+⑤A: 70.0 ng/ul 2.22; ⑧+⑤B: 35.6 ng/ul 2.84; ⑨+⑤A: 65.9 ng/ul 2.23; ⑨+⑤B: 74.3 ng/ul 2.18;
‘’’Comments:’’’ All the concentration seems to have some problem, and the A260/280 have some problem.
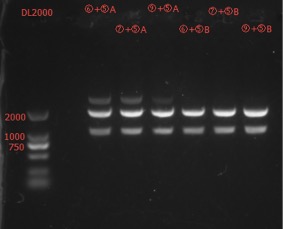
Enzyme Digestion and Go Gel Electrophoresis Test of ⑥+⑤, ⑦+⑤, ⑧+⑤, ⑨+⑤ A and B
Time: 23:00
Handler: Tang Shiqiang
Procedure:
- Make enzyme cutting system solution.
| ⑥+⑤A | ⑦+⑤A | ⑧+⑤A | ⑨+⑤A | ⑥+⑤B | ⑦+⑤B | ⑧+⑤B | ⑨+⑤B | |
|---|---|---|---|---|---|---|---|---|
| DNA(200ng) | 4ul | 4ul | 3ul | 3ul | 7ul | 4ul | 7ul | 3ul |
| Cutsmart buffer | 2ul | 2ul | 2ul | 2ul | 2ul | 2ul | 2ul | 2ul |
| XbaI | 0.2ul | 0.2ul | 0.2ul | 0.2ul | 0.2ul | 0.2ul | 0.2ul | 0.2ul |
| PstI-HF | 0.2ul | 0.2ul | 0.2ul | 0.2ul | 0.2ul | 0.2ul | 0.2ul | 0.2ul |
| ddH2O | 13.6ul | 13.6ul | 14.6ul | 14.6ul | 10.6ul | 13.6ul | 10.6ul | 14.6ul |
| Total | 20ul | 20ul | 20ul | 20ul | 20ul | 20ul | 20ul | 20ul |
- Incubate at 37℃ for 30 min.
- Make 1% agarose gel 30ml.
- Add 6x loading dye to the enzyme cutting system solution.
- Go gel electrophoresis.
Results:
The length seems to be correct.
8.5
Enzyme Digestion for Gel Extraction of ⑥+⑤, ⑦+⑤, ⑧+⑤, ⑨+⑤
Time: 2:20
Handler: Tang Shiqiang
Procedure:
- Make enzyme cutting system solution.
| ⑥+⑤A | ⑦+⑤B | ⑧+⑤A | ⑨+⑤B | |
|---|---|---|---|---|
| DNA(>1.5ug) | 26ul | 26ul | 26ul | 26ul |
| Cutsmart buffer | 3ul | 3ul | 3ul | 3ul |
| EcoRI-HF | 0.5ul | 0.5ul | 0.5ul | 0.5ul |
| XbaI | 0.5ul | 0.5ul | 0.5ul | 0.5ul |
| ddH2O | 0ul | 0ul | 0ul | 0ul |
| Total | 30ul | 30ul | 30ul | 30ul |
Incubate at 37℃ overnight. (Start from 2:30)
Go Gel Electrophoresis and Gel Extraction of ⑥+⑤, ⑦+⑤, ⑧+⑤, ⑨+⑤
Time: 8:10
Handler: Lu Shixin, Tang Shiqiang
Procedure: bio-tek OMEGA E.Z.N.A. Gel Extraction Kit – Spin Protocol
Results:
Concentration: ⑥+⑤: 25.1 ng/ul 1.58; ⑦+⑤: 23.4 ng/ul 1.54; ⑧+⑤: 45.9 ng/ul 1.01; ⑨+⑤: 26.7 ng/ul 1.50;
Ligation of ⑩,11 and ⑥+⑤, ⑦+⑤, ⑧+⑤, ⑨+⑤
Time: 11:00
Handler: Lu Shixin, Tang Shiqiang
Procedure:
- Make ligation system solution.
| ⑩+⑥+⑤ | ⑩+⑦+⑤ | ⑩+⑧+⑤ | ⑩+⑨+⑤ | |
|---|---|---|---|---|
| Vector ⑩ (50ng) | 0.8ul | 0.8ul | 0.8ul | 0.8ul |
| Insert | 3.4ul | 3.4ul | 2ul | 3.4ul |
| ddH2O | 5.8ul | 5.8ul | 7.2ul | 5.8ul |
| 2x Quick ligase buffer | 10ul | 10ul | 10ul | 10ul |
| Quick ligase | 1ul | 1ul | 1ul | 1ul |
| Total | 21ul | 21ul | 21ul | 21ul |
| 11+⑥+⑤ | 11+⑦+⑤ | 11+⑧+⑤ | 11+⑨+⑤ | |
|---|---|---|---|---|
| Vector ⑩ (50ng) | 0.8ul | 0.8ul | 0.8ul | 0.8ul |
| Insert | 3.4ul | 3.4ul | 2ul | 3.4ul |
| ddH2O | 5.8ul | 5.8ul | 7.2ul | 5.8ul |
| 2x Quick ligase buffer | 10ul | 10ul | 10ul | 10ul |
| Quick ligase | 1ul | 1ul | 1ul | 1ul |
| Total | 21ul | 21ul | 21ul | 21ul |
- Incubate at room temperature for 10 min.
- Take 5ul ligation system solution to do transformation.
8.6
Observation of single colonies of ⑩+⑥+⑤, ⑩+⑦+⑤, ⑩+⑧+⑤, ⑩+⑨+⑤, 11+⑥+⑤, 11+⑦+⑤, 11+⑧+⑤, 11+⑨+⑤
Time: 7:00
Handler: Xiao Tianyao
Results:
| Colonies number | |
|---|---|
| ⑩ backbone control | 5 |
| 11 backbone control | 20 |
| ⑩+⑥+⑤ | None |
| ⑩+⑦+⑤ | 1 |
| ⑩+⑧+⑤ | 20 |
| ⑩+⑨+⑤ | >50 |
| 11+⑥+⑤ | 4 |
| 11+⑦+⑤ | 1 |
| 11+⑧+⑤ | 6 |
| 11+⑨+⑤ | >50 |
Comments: Control groups have colonies. So the ligation results are improbable. We would redo it.
Picking single colonies of ⑩+⑦+⑤, ⑩+⑧+⑤, ⑩+⑨+⑤, 11+⑥+⑤, 11+⑦+⑤, 11+⑧+⑤, 11+⑨+⑤
Time: 10:00
Handler: Liao Weiduo
Procedure:
- Pick single colonies, adding 3 ml C+ LB.
- Incubate at 37℃ for about 12h, 220rpm.
Picking single colonies of ⑥+⑤, ⑦+⑤, ⑧+⑤, ⑨+⑤
Time: 11:00
Handler: Tang Shiqiang
Procedure:
- Pick single colonies, adding 6ml LB with corresponding antibiotics, each plate we pick 2 single colonies, C and D.
- Incubate at 37℃ for about 12h, 220rpm.
Plasmid extraction of ⑥+⑤, ⑦+⑤, ⑧+⑤, ⑨+⑤ A and B
Time: 23:00
Handler: Tang Shiqiang
Procedure: bio-tek OMEGA E.Z.N.A. Plasmid DNA Mini Kit I Spin Protocol
Results:
Concentration: ⑥+⑤C: 63.8 ng/ul 1.81; ⑥+⑤D: 59.4 ng/ul 1.79; ⑦+⑤C: 49.9 ng/ul 1.81; ⑦+⑤D: 65.8 ng/ul 1.75; ⑧+⑤C: 73.4ng/ul 1.81; ⑧+⑤D: 72.8 ng/ul 1.81; ⑨+⑤C: 65.8 ng/ul 1.74; ⑨+⑤D: 64.1 ng/ul 1.83.
8.7
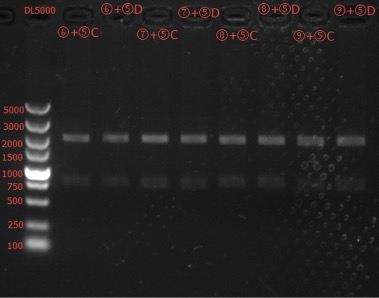
Enzyme Digestion and Go Gel Electrophoresis Test of ⑥+⑤, ⑦+⑤, ⑧+⑤, ⑨+⑤ C and D
Time: 2:00
Handler: Tang Shiqiang
Procedure:
- Make enzyme cutting system solution.
| ⑥+⑤C | ⑦+⑤C | ⑧+⑤C | ⑨+⑤C | ⑥+⑤D | ⑦+⑤D | ⑧+⑤D | ⑨+⑤D | |
|---|---|---|---|---|---|---|---|---|
| DNA(100ng) | 1.5ul | 1.5ul | 1.5ul | 1.5ul | 1.5ul | 1.5ul | 1.5ul | 1.5ul |
| Cutsmart buffer | 2ul | 2ul | 2ul | 2ul | 2ul | 2ul | 2ul | 2ul |
| XbaI | 0.2ul | 0.2ul | 0.2ul | 0.2ul | 0.2ul | 0.2ul | 0.2ul | 0.2ul |
| PstI-HF | 0.2ul | 0.2ul | 0.2ul | 0.2ul | 0.2ul | 0.2ul | 0.2ul | 0.2ul |
| ddH2O | 16.1ul | 16.1ul | 16.1ul | 16.1ul | 16.1ul | 16.1ul | 16.1ul | 1.16ul |
| Total | 20ul | 20ul | 20ul | 20ul | 20ul | 20ul | 20ul | 20ul |
- Incubate at 37℃ for 30 min.
- Make 1% agarose gel 30ml.
- Add 6x loading dye to the enzyme cutting system solution.
- Go gel electrophoresis.
Results:
Observation of LB medium color of ⑩+⑥+⑤, ⑩+⑦+⑤, ⑩+⑧+⑤, ⑩+⑨+⑤, 11+⑥+⑤, 11+⑦+⑤, 11+⑧+⑤, 11+⑨+⑤
Time: 7:00
Handler: Xiao Tianyao
Results:
| Color | |
|---|---|
| ⑩+⑥+⑤ | None |
| ⑩+⑦+⑤ | Purple |
| ⑩+⑧+⑤ | Yellow |
| ⑩+⑨+⑤ | Red |
| 11+⑥+⑤ | Red |
| 11+⑦+⑤ | None |
| 11+⑧+⑤ | None |
| 11+⑨+⑤ | None |
‘’’Note:’’’ There are yellow colonies on plate 11+⑧+⑤ and red colonies on 11+⑨+⑤.
Picking single colonies of ⑩+⑦+⑤, ⑩+⑧+⑤, ⑩+⑨+⑤, 11+⑥+⑤, 11+⑧+⑤, 11+⑨+⑤ (Because there are obvious correct color colonies on those plates)
Time: 9:30
Handler: Liao Weiduo
Procedure:
- Pick single colonies, adding 6 ml C+ LB.
- Incubate at 37℃ for about 12h, 220rpm.
Ligation of ⑩ and ⑥+⑤, 11 and ⑦+⑤
Time: 11:00
Handler: Tang Shiqiang
Procedure:
- Make ligation system solution.
| ⑩+⑥+⑤ | |
|---|---|
| Vector ⑩ (50ng) | 0.7ul |
| Insert | 3.2ul |
| ddH2O | 6.1ul |
| 2x Quick ligase buffer | 10ul |
| Quick ligase | 1ul |
| Total | 21ul |
| 11+⑦+⑤ | |
|---|---|
| Vector ⑩ (50ng) | 0.7ul |
| Insert | 3.2ul |
| ddH2O | 6.1ul |
| 2x Quick ligase buffer | 10ul |
| Quick ligase | 1ul |
| Total | 21ul |
- Incubate at room temperature for 10 min.
- Take 5ul ligation system solution to do transformation.
Plasmid extraction of ⑩+⑦+⑤, ⑩+⑧+⑤, ⑩+⑨+⑤, 11+⑥+⑤, 11+⑧+⑤, 11+⑨+⑤
Time: 22:00
Handler: Liao Weiduo
Procedure: bio-tek OMEGA E.Z.N.A. Plasmid DNA Mini Kit I Spin Protocol
Results:
Concentration: ⑩+⑦+⑤: 63.6 ng/ul 1.71; ⑩+⑧+⑤: 100.7 ng/ul 1.54; ⑩+⑨+⑤: 83.0 ng/ul 1.59; 11+⑥+⑤: 114.8 ng/ul 1.65; 11+⑧+⑤: 77.5 ng/ul 1.78; 11+⑨+⑤: 106.3 ng/ul 1.51.
8.9
Picking single colonies of ⑩+⑥+⑤, 11+⑦+⑤
Time: 00:00
Handler: Tang Shiqiang
Procedure:
- Pick single colonies, adding 6ml LB with corresponding antibiotics.
- Incubate overnight at 37℃, 220rpm.
Plasmid extraction of ⑩+⑥+⑤, 11+⑦+⑤
Time: 16:00
Handler: Tang Shiqiang
Procedure: bio-tek OMEGA E.Z.N.A. Plasmid DNA Mini Kit I Spin Protocol
Results:
Concentration: ⑩+⑥+⑤: 109.0 ng/ul 1.74; 11+⑦+⑤: 115.3 ng/ul 1.85.
sfGFP notebook
8.2
Plate streaking of a plasmid with contain sfGFP and msfGFP
Time: 17:30
Handler: Tang Shiqiang
8.3
Picking single colonies of sfGFP and msfGFP
Time: 9:00
Handler: Tang Shiqiang
Procedure:
- Pick single colonies, adding 6ml LB with corresponding antibiotics.
- Incubate for about 12h at 37℃, 220rpm.
Plasmid extraction of sfGFP and msfGFP
Time: 21:30
Handler: Lu Shixin
Procedure:bio-tek OMEGA E.Z.N.A. Plasmid DNA Mini Kit I Spin Protocol
Result:
Concentration: sfGFP: 70.0 ng/ul 1.77; msfGFP: 460.8 ng/ul.
8.16
PCR of sfGFP and msfGFP
Time: 16:00
Handler: Tang Shiqiang
Procedure:
| DNA | 0.1ul |
|---|---|
| bbp3 | 2.5ul |
| bbp4 | 2.5ul |
| Q5 | 25ul |
| ddH2O | 20ul |
| Total | 50ul |
| 32 cycles | Temperature | Time |
|---|---|---|
| Initialization | 98℃ | 30s |
| Denaturation | 98℃ | 5s |
| Annealing | 65℃ | 20s |
| Extension | 72℃ | 10s |
| Final elongation | 72℃ | 2min |
Go Gel Electrophoresis and Gel Extraction of sfGFP and msfGFP PCR product
Time: 19:00
Handler: Tang Shiqiang
Procedure: bio-tek OMEGA E.Z.N.A. Gel Extraction Kit – Spin Protocol
Result:
Concentration: sfGFP: 43.2 ng/ul 1.76; msfGFP: 62.0 ng/ul 1.79.
Enzyme Digestion for Gel Extraction of sfGFP and msfGFP
Time: 19:30
Handler: Tang Shiqiang
Procedure:
- Make enzyme cutting system solution.
| sfGFP | msfGFP | |
|---|---|---|
| DNA(>1ug) | 27.5ul | 27.5ul |
| Cutsmart buffer | 2.5ul | 2.5ul |
| EcoRI-HF | 0.5ul | 0.5ul |
| SpeI-HF | 0.5ul | 0.5ul |
| ddH2O | 19ul | 19ul |
| Total | 50ul | 50ul |
- Incubate at 37℃ for 3h.
Enzyme Digestion for Gel Extraction of ⑤
Time: 17:00
Handler: Tang Shiqiang
Procedure:
- Make enzyme cutting system solution.
| ⑤ | |
|---|---|
| DNA(>3ug) | 21.5ul |
| Cutsmart buffer | 2.5ul |
| EcoRI-HF | 0.5ul |
| SpeI-HF | 0.5ul |
| ddH2O | 0 |
| Total | 25ul |
- Incubate at 37℃ for 3h
Go Gel Electrophoresis and Gel Extraction of sfGFP, msfGFP, and ⑤
Time: 20:00
Handler: Tang Shiqiang
Procedure: bio-tek OMEGA E.Z.N.A. Gel Extraction Kit – Spin Protocol
Results:
Gel extraction concentration: ⑤: 61.4 ng/ul 1.58; sfGFP: 16.9 ng/ul 1.62; msfGFP: 16.5 ng/ul 1.59.
8.17
Ligation of sfGFP+⑤, msfGFP+⑤, sfGFP+pSB1C3, msfGFP+pSB1C3
Time: 0:00
Handler: Tang Shiqiang
Procedure:
- Make ligation system solution.
| sfGFP | msfGFP | |
|---|---|---|
| Vector ⑤ (50ng) | 1ul | 1ul |
| Insert | 4ul | 4ul |
| ddH2O | 0ul | 0ul |
| 2x Quick ligase buffer | 5ul | 5ul |
| Quick ligase | 0.5ul | 0.5ul |
| Total | 10.5ul | 10.5ul |
- Incubate at room temperature for 10 min.
- Take 10.5ul ligation system solution to do transformation.
Picking single colonies of sfGFP and msfGFP
Time: 18:30
Handler: Tang Shiqiang
Procedure:
- Pick single colonies, adding 6ml LB with corresponding antibiotics.
- Incubate overnight at 37℃, 220rpm.
8.18
Plasmid extraction of sfGFP+⑤, msfGFP+⑤, sfGFP+pSB1C3, msfGFP+pSB1C3
Time: 13:00
Handler: Tang Shiqiang
Procedure: bio-tek OMEGA E.Z.N.A. Plasmid DNA Mini Kit I Spin Protocol
Result:
Concentration: sfGFP+⑤: 103.6 ng/ul 1.96; msfGFP+⑤: 127.1 ng/ul 1.80; sfGFP+pSB1C3①: 156.1 ng/ul 1.91; sfGFP+pSB1C3②: 179.2 ng/ul 1.91; msfGFP+pSB1C3①: 115.5 ng/ul 1.93; msfGFP+pSB1C3②: 159.5 ng/ul 1.92.
8.18
Enzyme Digestion and Go Gel Electrophoresis Test of sfGFP+⑤, msfGFP+⑤, sfGFP+pSB1C3, msfGFP+pSB1C3
Time: 0:00
Handler: Tang Shiqiang
Procedure:
- Make enzyme cutting system solution.
| sfGFP+⑤ | msfGFP+⑤ | sfGFP+pSB1C3① | sfGFP+pSB1C3② | msfGFP+pSB1C3① | msfGFP+pSB1C3② | |
|---|---|---|---|---|---|---|
| DNA(200ng) | 2ul | 2ul | 2ul | 2ul | 2ul | 2ul |
| Cutsmart buffer | 2ul | 2ul | 2ul | 2ul | 2ul | 2ul |
| KpnI-HF | 0.2ul | 0.2ul | 0.2ul | 0.2ul | 0.2ul | 0.2ul |
| ddH2O | 16.8ul | 16.8ul | 16.8ul | 16.8ul | 16.8ul | 16.8ul |
| Total | 20ul | 20ul | 20ul | 20ul | 20ul | 20ul |
| sfGFP+⑤ | msfGFP+⑤ | sfGFP+pSB1C3① | sfGFP+pSB1C3② | msfGFP+pSB1C3① | msfGFP+pSB1C3② | |
|---|---|---|---|---|---|---|
| DNA(200ng) | 2ul | 2ul | 2ul | 2ul | 2ul | 2ul |
| Cutsmart buffer | 2ul | 2ul | 2ul | 2ul | 2ul | 2ul |
| EcoRI-HF | 0.2ul | 0.2ul | 0.2ul | 0.2ul | 0.2ul | 0.2ul |
| ddH2O | 16.8ul | 16.8ul | 16.8ul | 16.8ul | 16.8ul | 16.8ul |
| Total | 20ul | 20ul | 20ul | 20ul | 20ul | 20ul |
| sfGFP+⑤ | msfGFP+⑤ | sfGFP+pSB1C3① | sfGFP+pSB1C3② | msfGFP+pSB1C3① | msfGFP+pSB1C3② | |
|---|---|---|---|---|---|---|
| DNA(200ng) | 2ul | 2ul | 2ul | 2ul | 2ul | 2ul |
| Cutsmart buffer | 2ul | 2ul | 2ul | 2ul | 2ul | 2ul |
| EcoRI-HF | 0.2ul | 0.2ul | 0.2ul | 0.2ul | 0.2ul | 0.2ul |
| PstI-HF | 0.2ul | 0.2ul | 0.2ul | 0.2ul | 0.2ul | 0.2ul |
| ddH2O | 16.6ul | 16.6ul | 16.6ul | 16.6ul | 16.6ul | 16.6ul |
| Total | 20ul | 20ul | 20ul | 20ul | 20ul | 20ul |
- Incubate overnight at 37℃.
- Make 1% agarose gel 30ml.
- Add 6x loading dye to the enzyme cutting system solution.
- Go gel electrophoresis.
Results:
9.21
PCR of msfGFP Coding sequence basic part
Time: 16:00
Handler: Tang Shiqiang
Procedure:
| DNA | 1ul |
|---|---|
| SfGFP-forward | 1ul |
| bbp4 | 1ul |
| primeSTAR | 20ul |
| ddH2O | 17ul |
| Total | 40ul |
| 32 cycles | Temperature | Time |
|---|---|---|
| Initialization | 98℃ | 30s |
| Denaturation | 98℃ | 10s |
| Annealing | 65℃ | 5s |
| Extension | 72℃ | 5s |
| Final elongation | 72℃ | 2min |
Go Gel Electrophoresis and Gel Extraction of msfGFP CDS
Time: 20:00
Handler: Tang Shiqiang
Procedure: bio-tek OMEGA E.Z.N.A. Gel Extraction Kit – Spin Protocol
Result:
Concentration: msfGFP: 96.1 ng/ul 1.83.
Enzyme Digestion for Gel Extraction of msfGFP CDS
Time: 20:30
Handler: Tang Shiqiang
Procedure:
- Make enzyme cutting system solution.
| msfGFP CDS | |
|---|---|
| DNA | 43ul |
| Cutsmart buffer | 5ul |
| EcoRI-HF | 1ul |
| SpeI-HF | 1ul |
| ddH2O | 0ul |
| Total | 50ul |
- Incubate overnight at 37℃.
9.22
Transformation of msfGFP CDS
Time: 11:00
Handler: Tang Shiqiang
Procedure: IGEM Protocols
Picking single colonies of sfGFP CDS
Time: 23:00
Handler: Tang Shiqiang
Procedure:
- Pick single colonies, adding 5ml C+ LB.
- Incubate overnight at 37℃, 220rpm.
9.23
Plasmid extraction of msfGFP CDS
Time: 17:00
Handler: Tang Shiqiang
Procedure: bio-tek OMEGA E.Z.N.A. Plasmid DNA Mini Kit I Spin Protocol
Result:
Concentration: msfGFP CDS①: 141.0 ng/ul 1.88; msfGFP CDS②: 141.0 ng/ul 1.90.
9.24
Enzyme Digestion and Go Gel Electrophoresis Test of msfGFP CDS① and msfGFP CDS②
Time: 11:40
Handler: Tang Shiqiang
Procedure:
- Make enzyme cutting system solution.
| msfGFP CDS① | msfGFP CDS② | |
|---|---|---|
| DNA(300ng) | 2.5ul | 2.5ul |
| Cutsmart buffer | 1ul | 1ul |
| MluI-HF/MscI | 0.5ul | 0.5ul |
| ddH2O | 6ul | 6ul |
| Total | 10ul | 10ul |
- Incubate for 3h at 37℃.
- Make 1% agarose gel 30ml.
- Add 6x loading dye to the enzyme cutting system solution.
- Go gel electrophoresis.
Results: