6/13/16
(HC) Made LB-Amp plates
450ml of DI water
5.0g tryptone
2.5g yeast
5.0g NaCl
7.5g Agar
This was then adjusted to a pH of 7.0 using NaOH.
It was then autoclaved. 2.5mL of 10mg/mL of Amp
for a final concentration of 50 μg/mL was achieved.
6/14/16
(HC) made CSM -His plates
6/15/16
(XX) Goal: Run a gel to check whether pSB416 GPD has PstI site removed (diagnostic gel)
1. pSB416 GPD cut wit AvaI
2. pSB416 GPD cut with PstI
6 sterile water, 1 CutSmart buffer, 2 DNA, 1 enzyme (AvaI or PstI-HF)
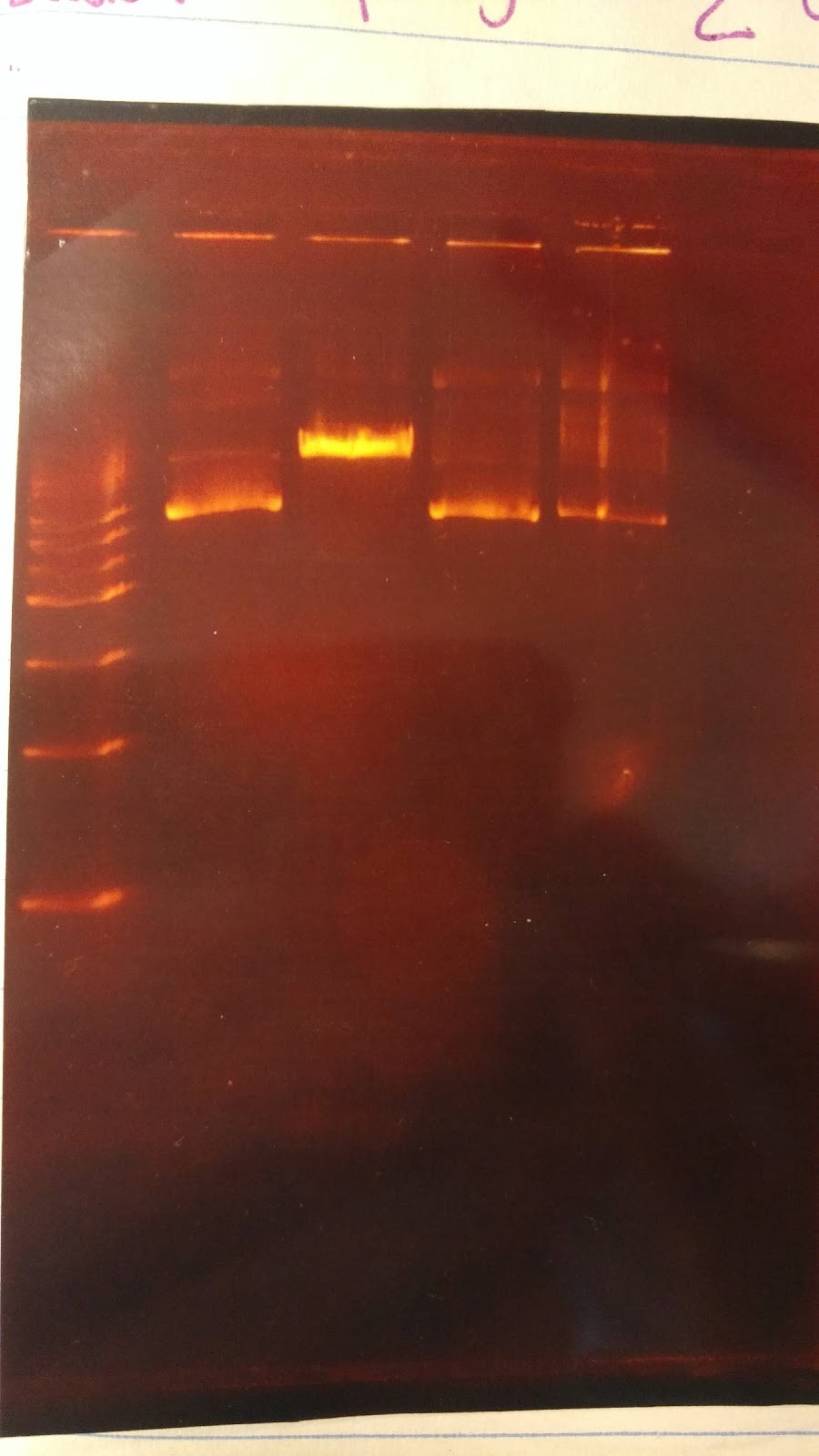
Expected results on this diagnostic gel:
1. No cut because no AvaI site in pSB416 GPD
2. Should only see one cut because only one PstI site
Created mRPS12+/KanMZ- (parent) and mRPS12-/KanMX+ (mutant) master plate on YPD
Replica plating YPG (Should this be YPD???) , YPG, -His, -Ura, -Ura+Glycerol, YPD+Paro, YPG+Paro
6/16/16
(HC) Ran a boiling and spin prep on P413 GPD
1) Pellet cells from 1.5mL of an overnight L-Amp culture of transformed E. coli cells in a microcentrifuge for 20-30 seconds and remove supernatant.
2) Use a toothpick and vortexer to resuspend the cells in 100 μl(0.1mL) of STET buffer(8% sucrose; 5%Triton X-100;50 mM EDTA; Tris-Cl, pH 8.0) containing 1mg/mL lysozyme(prepare 1 mg/mLenzyme in STET just prior to use; 1mL= 10 preps).
3)Place the suspension in a boiling water bath for 90 seconds.
4) Spin in a microcentrifuge for 15 min at the highest speed.
5)Remove the pellet (a gelatinous mass of cell debris) with a toothpick and discard.
6) Precipitate nucleic acid at -20℃ for 30 minutes using an equal volume of isopropanol.
7) Spin the tube in a microcentrifuge for 10 minutes at RTo to pellet the nucleic acids.
8) Was pellet twice with 70% ethanol and dry thoroughly.
9) Resuspend the pellet in 50 μL of sterile DI water or TE buffer.
Spin Prep: We used the QIAprep Spin Miniprep Kit to run a spin prep and followed the instructions inside.
(XX)Goal: Rerun the diagnostic gel stated yesterday (6/15/16)
Good gel results, confirming that the PstI site has been removed in pSB416 GPD. pSB416 ready to be sequenced.
Incubated pSBIC3 mRPS12 TU and pSBIC3 mRPS12 mls (from last year iGEM team??) in LB-chloramphenicol broth @37C overnight at 250 rpm
6/17/16
(HC) Ran two digests for P413 GPD
5 μL of DNA(Boiling Prep)
Lane 3: Preparative Digest
INSERT NOTEBOOK PICTURE #001 HERE
ANALYTICAL GEL
Lane 2: Uncut (2μL of DNA and 8μL of water)
Lane 3: Analytical Digest
INSERT NOTEBOOK PICTURE #002 HERE
(XX) Master plate results:
Nothing grew on control plates (-His, -Ura, -Ura+Glycerol)
Both parent and mutant grew on YPD and YPD+Paro, indicating that Paro has no effect on yeast growth
Mutant cannot grow on YPG or YPG+Paro because of the mRPS12 gene knockout
Cut p413GPD with NheI and NsiI to get rid of the illegal PstI site
Massive gel to run boiling preps
P413GPD cut looks like an uncut plasmid
Massive gel has many blurry bands, which indicates that boiling preps are very impure
6/20/16
Put pSBIC3 mRPS12 TU into our own standardized yeast vectors
Validate mls function by creating mls-yeGFP construct
Digest and Harvest mRPS12 TU from pSBIC3(tube 1)
Digest and Harvest mRPS12 mls from pSBIC3 (tube 2)
Gel Order: L, 1C, 1XX, 1XL, 2C, 2XX, 2XL
**C-uncut plasmid, XX-Xintong, XL-Xander
Uncut does not show up on the gel
Not a very good results but we harvested the bands anyway
Excise bands from the gel and perform gel extraction (QIA kit):
*tube 3 contains p413 GPD with NsII/NheI cut
Prepare Yeast Competent Cells
10 mL YPD, inoculated at 250RPM @ 30C
Both BY4741 and BY4741 mRPS12 is prepared
6/21/16
We used nanodrop to determine the amount of DNA in tube 1 (TU) and tube 2 (mls)
* Nano drop results indicate that we need to precipitate DNA for further experiment
After precipitation, nanodrop reports 12.6 ng/ DNA
NEBuilder HiFi DNA Assembly Cloning Kit:
2μL Ear/SpeI digest pSBIC3 mls
8μL fragment 16-4 (yeGFP)
*All three above, set the reaction on ice; 1:2 vecotr: insert
Yeast competent cell from yesterday did not grow
Prepare new yeast competent cells: 5 mL YPD in round bottom tubes
6/22/16
Prepare Yeast Competent cells
Diagnostic gel on mRPS12 TU minipreps
Spectromphotometer OD 260:
1000 KnockOut(KO) has 1.777 Abs.
1000 Parent has 2.222 Abs.
Regrow these cells into 15 mL broth for about 2 hrs:
Take 0.5 mL current culture into 14.5 mL YPD
Spectrophotometer Results:
Follow yeast competent cell protocol
1: pSBIC3 mRPS12 TU miniprep 1 cut with EcoRI and PstI
2: pSBIC3 mRPS12 TU miniprep 2 cut with EcoRI and PstI
3: pSBIC3 mRPS12 TU miniprep 1 cut with PstI
4: pSBIC3 mRPS12 TU miniprep 2 cut with PstI
For #2, fragment should be around 500bp
Gel results indicate that we should redo minipreps or bad enzymes
6/23/16
Test if the enzymes are still okay to work with
Redo miniprep at the same time
Gel Results: enzymes are working properly
6/24/16
(HC) After extracting P413 GPD fragment from the gel, I precipitated the DNA
1.)Add 1-2 μL of 5M NaCl to 30 μL of DNA
2.)Add 62 μL of ice cold EtOH
3.)Let sit on ice for 30 minutes
4.)Centrifuge @ 0℃ for 10 minutes at 13 krpm
6.)Fill tube to halfway point with 70% EtOH
7.)Centrifuge @ 4℃ for 2 minutes
10.)Add 6μL of Sterile Water.
Obtained concentration of DNA with NanoDropper
| Nucleic Acid Concentratione |
A @ 260 nm |
A @ 280 nm |
260/280 |
260/230 |
| 75.7 ng/μL |
1.514 |
0.922 |
1.64 |
.033 |
Today we are starting the day off by running a gel.
We just called iGEM to confirm that we can indeed submit vectors as parts
The gel we are running today is to confirm that we only have 1 PstI site in all of our plasmids
The hope: to see one solid band from all of our plasmids except for the controls, the controls should be two bands because they still have two PstI sites
Another thing we are doing today is we are making media LB Chloro.
| 1 |
2 |
3 |
4 |
5 |
6 |
| 416 control |
426 control |
416 CYC1 |
416 CYC1 |
426 GPD |
426 GPD |
(XX) Talked about light switch design
LexA promoter instread of Gal promoter
PhyB/PIF3 red light system
6/25/16
(XX) Miniprep mRPS12 TU and mRSP12 mls(total of 8 minipreps)
6/27/16
(XX) Goal: Redo digest and harvest TU and mls
Cut mRPS12 TU with EcoRI/PstI
Cut Mrps12 mls with EarI/SpeI
Gel order: L,TU1a, TU1b, TU2a, TU2b, mls1a, mls1b, mls2a, mls2b
harvested TU bands from TU2a and TU2b (2 bands per tube)
harvested four mls bands (2 bands per tube)
mls gel extract A = 0.342 g
mls gel extract B = 0.255 g
Used nanodrop to test DNA amount:
*according to nanodrop readings, TU needs to be precipitate while we run NEBuilder on mls A and mls B
(HC) Ran NEBuilder on P413 GPD and 16-1 fragment.
10μL NEBuilder HiFi DNA Assembily Master Mix
The first thing we are doing is running the blunt end ligation protocol using NEB Quick Blunting Kit and the NEB Quick Ligation Kit
This was done with 3 tubes of 416 CYC1 and 3 tubes of 426 GPD
Next we did a transformation on one of the 416 CYC1 and one of the 426 GPD
Those are spread on plates and are now incubating so we can use them tomorrow
6/28/16
Slow day today because all we can do is inoculate falcon tubes for 24 hours so we can run a mini-prep tomorrow
We did 6 tubes of 416 CYC1
perform transformation for TU, mls A, and mls B
TU1a, TU2a: just PstI enzyme
TU1b, TU2b: double digest (PstI/EcoRI)
Redo NEBuilder mls A and mls B
*due to programing issue??
Gel order: L, Uncut (TU), PstI TU1a, PstI/EcoRI TU1b, PstI TU2a, PstI/EcoRI TU2b
the band after only PstI cut seems too long – questioning the pSBIC3 mRPS12 TU stock??
6/29/16
After incubating for an undetermined amount of time due to our shaking incubators being complete pieces of shit we are now going to mini preps because we could not find the proper buffer for the boiling preps
With the miniprep material we had we were able to do 3 tubes of 416 and 2 tubes of 426
Ran the digest of the plasmids with Pst1-HF
(XX) Transformation results: No transformants for mls
Cut with only EarI and run gel
Streak pRS416 (the vector with no promoter) on LB-Amp plates – for LexA light switch
Transform competent E. Coli with pSBGPD416 A, plate on LB-Amp
Gel order: L, mls A uncut, mls 1a cut
Gel results: bad mls miniprep – questioning the mls stock??
6/30/16
(JM/CH)
Today we are running the gel of the digest that should result in seeing only one Pst1 site
Gel Set-up
BIG OL TABLE GOES HERE
Results: too blurry to come to any reasonable conclusion
Now to set up a proper gel to run tomorrow
We need to set up two controls, which will be minipreps of the DNA plasmids with 2 Pst1 sites
Also we will be using 5 of our blunt end ligated DNA (3 416, and 2 426)
We will then also load two of the wells, one with 416 and one with 426, but uncut
(XX) Found out that pSB416GPD I from last year has failed because it has an extra start codon before BB prefix
Goal: extract mRPS12 TU from old pSB416 GPD I
Major steps:
Made a 20 digest – 10 to run gel and 10 to run NEBuilder
10 pSB416 GPD I from last year
1 EcoRI
1 PstI
2 CutSmart Buffer
7 sterile water
Other activities:
Streak pSBIC3mls I on LB-chloro
Inoculate pSB416 GPD A in LB-Amp
Inoculate pRS416 in LB-Amp
Gel Results:
Nice mRPS12 TU band on the gel, extract mRPS12 TU band
Mass = 0.265 g
Nanodrop results TU: 5.6 ng/
*need to do precipitation
7/1/16
(HC) Ran a spin miniprep using the Monarch Nucleic Acid Purification Kit and the instructions inside on the P413 Transformant.
Ran a digest
7μL of DNA
1μL of Cutsmart
1μL of PstI
1μL of Sterile water
Ran gel with cut and uncut transformant
Lane1: Ladder
Lane 2: Cut
Lane 3: Uncut
Lane1: Ladder
Lane 2: Cut
Lane 3: Uncut
(JM/CH)
Today we ran the gel that had out plasmids hopefully set for perfection
ladder 416 1 416 2 416 3 416 orig 416 uncut 426 uncut 426 orig 426 1 426 2
Results:
Yayayayayay. This shows that our gel yesterday is confirmed and every sample shows one band. The Pst1 worked due to the double bands in wells 5 and 8. Edit: After talk with PI the gel does not have correct size of bands. Still inconclusive.
(XX) Precipitation of TU from yesterday resulted 3.8 ng/ according to nanodrop. This result does not make sense and it might possibly due to nanodrop defecting DNA after gel isolation. So we continue on NEBuilder anyway.
Other activities:
miniprep pRS416 and pSB416 GPD A
inoculate pSBIC3 mls in LB-chloro broth
7/2/16
(XX) Goal:
Miniprep pSBIC3 mls
TU transformation
7/3/16
(XX)We see transformants on 7/3/16! This is exciting news!
7/4/16
7/5/16
(HC) Ran a digest with P413 GPD
2 μL of DNA
1μL of Cutsmart
1μL of PstI
6μL of Sterile Water
Ran Gel
*** Ran a couple different experiments on one gel and will only list the ones related to P413 GPD experiment, the others will be listed in another Lab members notebook section
Lane 1: Ladder
Lane 2: Cut
Lane 3: Uncut
Ran Digest on P413 GPD on two different preps
*Prep was run by Connor
#Prep was run by Holly
Both Preps were run through the same digests
2 μL of DNA
1 μL of NheI
1 μL of 2.1 buffer
6 μL of Sterile Water
2 μL of DNA
1 μL of NsiI
1 μL of Cutsmart buffer
6 μL of Sterile Water
2 μL of DNA
1 μL of PstI
1 μL of Cutsmart buffer
6 μL of Sterile Water
2 μL of DNA
1 μL of EARI
1 μL of cutsmart buffer
6 μL of Sterile Water
(XX) Goal:
Diagnostic gel of pSB416 GPD mRPS12 TU: used HindIII or PstI enzymes
Isolate mls DNA from mRPS12 mls miniprep: double digest using EarI and SpeI
Gel Order:
L, pSBIC3 Cut, pSBIC3 Uncut, B, mls digest, mls uncut, B, TU PstI, TU HindIII, TU uncut
Gel Results:
TU with HindIII has weird result, possibly due to star activity or failed digest?
Redo pSBIC3
Redo mls
Redo TU digest
Other activities:
Made master plate on CSM-D-URA
pSB416 GPD A mRPS12 (5-8)
pSB416 GPD A Parent (1-4)
7/6/16
(HC) Ran Gel
Lane1: Ladder, Lane2: NheI*, Lane 3: NheI#, Lane 4: NsiI*, Lane 5: NsiI#, Lane 6: PstI*,
Lane 7: PstI#, Lane 8: Ear1*, Lane 9: Ear1#, Lane 10: Uncut
Ran digest on P413 GPD of Holly’s prep(# prep from above)
2 μL of DNA
1 μL of EarI
1 μL of Cutsmart buffer
6 μL of Sterile Water
2 μL of DNA
1 μL of XhoI
1 μL of Cutsmart buffer
6 μL of Sterile Water
2 μL of DNA
1 μL of PstI
1 μL of Cutsmart buffer
6 μL of Sterile Water
Ran gel of these digests
Lane 1: Ladder, Lane 2: Uncut, Lane 3: Ear1, Lane 4: PstI, Lane 5: XhoI
7/7/16
(HC) Ran digest w/ EarI and XhoI on my preps for P413 GPD(*) and a team member’s prep of P413 GPD(#) from last year.
2 μL of DNA
1 μL of EarI
1 μL of Cutsmart buffer
6 μL of Sterile Water
2 μL of DNA
1 μL of XhoI
1 μL of Cutsmart buffer
6 μL of Sterile Water
Ran a gel to compare the preps of P413 GPD and the effects of Pre-staining vs. Post-Staining.
Gel 1: Pre-stained
Lane 1: 500bp Ladder, Lane 2: XhoI #, Lane 3: XhoI*, Lane 4: EarI#, Lane 5: EarI*
Gel 2: Not stained( we hadn’t stained this gel yet but it was clear to see bands)
Lane 1: 1kbp Ladder, Lane 2: XhoI #, Lane 3: XhoI*, Lane 4: EarI#, Lane 5: EarI*
(JM/CH)
Inoculated tubes that we will use tomorrow with 416 CYC1 and 426 GPD
Made up 50mL STET buffer
• 4g 8% Sucrose
• 2.5 mL 5% Trition X-100
• 5 mL 50 mM EDTA
• 2.5 mL 50 mM Tris-Cl
•
7/8/16
(JM)
Alpha and beta are different designations to distinguish between two possible candidates for testing.
ladder 416 pst1 alpha 416 EcoR1 alpha 416 Uncut alpha 416 Uncut beta 416 Pst1 beta 416 EcoR1 beta
ladder 426 pst1 alpha 426 EcoR1 alpha 426 Uncut alpha 426 Uncut beta 426 Pst1 beta 426 ecoR1 beta
Chose Both 416CYC1 alpha and 426 GPD alpha to freeze away and make new cryo tubes
(XX)Goal:
Redo pSB416 GPD mRPS12 TU diagnostic gel
Double digest pSBIC3 mRPS12 mls
Gel Order: L, TU, B, mls uncut, mls cut
Gel results:
TU has no insert, need to restart from NEBuilder
Can isolate mls cut band and do gel extraction
Other activities:
NEBuilder to form mls-yeGFP construct:
1 Digest
9 16-4 fragment
**Check for time-saver qualified enzymes – only need to digest for 5 to 15 minutes
7/17/16
(HC) Made Amp plates and CSM-URA plates
7/18/16
(HC) Plated P413 GPD onto AMP-Plates to allow to grow for colonies.
(JM/CH)
Today we are going to run two gels to test if prestaining or post staining is more effective
The gel inside the clear container while the nonstained one is in the blue one
Ladder 416 CYC1 cut w/ Pst1 416 CYC1 cut w/ EcoR1 416 CYC1 Uncut mRPS12 tu invalid Ladder #2
The gel on the right was not stained in any manner, but bands can still be easily seen.
(XX) Goal: Transformation for pSBIC3 mls-yeGFP on LB-Amp plate
7/19/16
(HC) Inoculated LB Amp tubes with colonies from plates
I selected two colonies from each plate and labeled them 1A, 1B, 2A, and 2B based on which plate and colony it was.
(JM/CH)
Today the plates have grown up nicely. We picked 3 colonies from each of the 2 plates (one 416 and one 426).
Now they are growing up in the incubator.
Creating lesson plan.
(XX) Transformation from yesterday did not work because of the LB-Amp plates!
**pSBIC3 is chloro selective!!
Goal: Plate NEBuilder stuff from 7/8/16 on LB-chloro plates
7/20/16
(HC)Ran a miniprep on all 4 tubes that were inoculated with the cells from the P413 GPD plates, 3 minipreps for each tube for a total of 12 tubes. I used the Monarch Plasmid Miniprep Kit.
Digested 1A, 1B, 2A, and 2B with NheI and NsiI
5 μL of DNA
1 μL of NheI
1 μL of NsiI
1 μL of Cutsmart buffer
2 μL of Sterile Water
I ran a gel with these digests.
Lane1: 1kbp Ladder, Lane 2: 1A uncut, Lane 3: 1A cut, Lane 4: 1B uncut, Lane 5: 1B cut,
Lane 6: 2A uncut, Lane 7: 2A cut, Lane 8: 2B uncut, Lane 9: 2B Cut
I cut out the bottom bands of Lanes 3, 5, 7, and 9 and kept them to eventually do a gel extraction.
(JM/CH)
Today we started off the day by doing mini preps to isolate our plasmids. Helped Holly with her 413 GPD miniprep. Worked on the 416CYC1 and 426 GPD miniprep
Now we are going to do the blunt end ligation to cut out the extra Pst1 site in both the 416CYC1 and 426 GPD plasmids.
The gel came back from the miniprep of 413 and can be seen above in Holly’s entry.
Now we are finishing the blunt end ligation of the 416 and 426. We kept tubes of just the blunted DNA and we will have a stock of the ligated DNA as well.
(XX) There are still no transformants for mls-yeGFP! L
Goal:
Miniprep TU transformants and diagnostic gel to check if we have any insert
Redo mRPS12 mls double digest – start mls-yeGFP process from gel extraction
Gel mass = 0.286 g
Gel Order:
L, A1, A2, A3, A4, B1, B2, Blank, mls uncut, mls cut
(A1 to B2) are minipreps from TU transformants
Gel Results: Gel shows TU transformants have no insert
7/21/16
(HC) Reran the digest because I cut out the wrong bands
Digested 1A, 1B, 2A, and 2B with NheI and NsiI
5 μL of DNA
1 μL of NheI
1 μL of NsiI
1 μL of Cutsmart buffer
2 μL of Sterile Water
(JM/CH)
The first thing we are going to do is run a digestion reaction with the enzyme Pst1
Today we ran a giant gel with 20 wells.
Wells 1-10:
Ladder 416B-1 cut 416-1 cut Uncut 416B-1 416B-2 cut 416-2 cut 416B-2 uncut 416B-3 cut 416-3 cut 416B-3 uncut
Wells 11-20:
426B-1 cut 426-1 cut 426B-1 uncut 426B-2 cut 426-2 cut 426B-2 uncut 426B-3 cut 426-3 cut 426B-3 uncut Ladder#2
Key:
B- blunted and ligated DNA
1,2,3- designation of different colonies
(XX)
Nanodrop pSBIC3 mRPS12 mls gel extract resulted 32.9 ng/
Goal:
NEBuilder to form mls-yeGFP
Transformation mls-yeGFP
Redo TU digest and harvest TU
Major steps:
NEBuilder (on ice):
2 mls digest
8 16-4 fragment
10 hifi master
Thermocycler 15 min. and store @ -20C
Transformation: plate on LB-chloro
TU gel extraction:
pSB416 GPD EcoRI/PstI: 47.9 ng/ (linearized plasmid)
TU digest: 11.3 ng/
7/22/16
(HC)
Ran the gel of the digest from the day before
Lane1: 1kbp Ladder, Lane 2: 1A uncut, Lane 3: 1A cut, Lane 4: 1B uncut, Lane 5: 1B cut,
Lane 6: 2A uncut, Lane 7: 2A cut, Lane 8: 2B uncut, Lane 9: 2B Cut
(INSERT PICTURE HERE)
Extracted from the gel lane 5 and Lane 7
Ran gel extraction from the monach gel Extraction kit.
Precipitated the DNA
1. Add 1-2 μL of 5M NaCl to 30μL of DNA
2. Add 62μL of ice cold EtOH
3. Let sit on ice for 30 minutes
4. Centrifuge @ 0℃ for 10 minutes @ 13 krpm
5. Remove supernatant
6. Fill tube halfway with 70% EtOH
7. Centrifuge @ 4℃ for 2 minutes
8. Remove supernatant and allow to dry
9. Add 6μL of water
Ran nano drop
Nucleic Acid Concentration A @ 260 nm A @ 280 nm 260/280 260/230
1.5 ng/μL 0.031 0.024 1.28 0.01
2.6 ng/μL 0.052 0.011 4.53 0.00
(XX)
mls-yeGFP transformants grow on plates!! J
pick 3 colonies from each plate and incubate the chosen colonies in LB-chloro broth overnight
7/23/16
(XX) Miniprep pSBIC3 mls-yeGFP:
mls 1 to mls 6
7/24/16
(XX)Goal: Diagnostic gel to test whether mls-yeGFP has insert
mls is small and will run off the gel, but mls-yeGFP can be detected; should see two bands if successfully inserted
Gel Order: L, mls 1, mls 2, mls 3, Blank, A1, B1
Gel results: minipreps mls 3, A1, and B1 seems to work
7/25/16
(HC)Ran NEBuilder with
1μL of DNA
1μL of Fragment 16-3
10μL of Builder Master Mix
8μL of Sterile Water
Made chemically competent Cells
Plated competent Cells
(JM/CH)
Today we are going to do boiling preps for the analytical digest today to verify if the Pst1 site has been properly cut out.
The boiling preps were done with 4 tubes of 416 CYC1 and 5 tubes of 426 GPD.
(XX) Goal:
Continue on diagnostic gel to test mls 4, 5, and 6
Quick ligation to insert mls-yeGFP into pSB416 GPD plasmid
Transformation
Major steps:
Mls-yeGFP digest:
5 DNA
1 EcoRI-HF, 1 PstI-HF
1 CutSmart Buffer
2 sterile water
Gel order: L, mls 4, mls 5, mls 6, Blank, A2, A3, B2
Gel Results: mls 4, mls 5, mls 6 and A2 seem working!!
Other activities:
Gel extraction mls-yeGFP DNA from gel:
mls 4 = 0.185 g
mls 5 = 0.166 g
mls 6 = 0.216 g
Nanodrop DNA reading:
mls 4: 17.8 ng/
mls 5: 14.6 ng/
mls 6: 16.7 ng/
Quick Ligation:
1.5 EcoRI/PstI Cut pSB416 GPD (vector)
1 mRPS12 mls-yeGFP (insert)
Follow NEB Quick ligation protocol online
*Do NOT heat inactivate!!
Transformation mls 4, mls 5, mls 6, A2 (transform TU miniprep back into pSB416 in order to make a stock)
7/26/16
(HC)Inoculated Cultures of LB Amp with selected cells
(INSERT PICTURE HERE)
(JM/CH)
Today is the day we find out if the blunt end ligation has worked. We are going to be running a digest and gel verification.
ladder b416 pst1 b416 pst1 b416 pst1 b416 pst1 b416 pst1 416 pst1 416 pst1 416 pst1 Uncut b416 Uncut b416 Uncut b416 Uncut b416 Uncut b416 ladder
ladder b426 pst1 b426 pst1 b426 pst1 b426 pst1 426 pst1 426 pst1 426 pst1 Uncut b426 Uncut b426 Uncut b426 Uncut b426 ladder
Any strain with a "b" in front of it means that it has gone through blunt end ligation.
(XX) Goal:
Nanodrop original mls 4, mls 5, and mls 6 minipreps
Inoculate culture
Nanodrop readings:
mls 4 = 184.2 ng/
mls 5 = 125.4 ng/
mls 6 = 163.6 ng/
7/27/16
(HC) Spinprep kit didn’t come in so I re-inoculated cultures in LB Amp
(JM/CH)
Today we are going to rerun the gel to try and get better results, apparently the boiling preps need to be centrifuged for ~15 min for better results.
ladder b416 pst1 b416 pst1 416 pst1 Uncut b416 Uncut b416 b426 pst1 b426 pst1 426 pst1 Uncut b426 Uncut b426 ladder
Any strain with a "b" in front of it means that it has gone through blunt end ligation.
(XX)Goal:
Dilute those DNA down to 100 ng/ for sequencing
Regrow culture
Major steps:
Miniprep volume left – about 24
Add 12 sterile water into A2, mls 4, mls 6
Add 6 sterile water into mls 5
Nanodrop readings after dilution:
A2: 41.4 ng/
mls 4: 94.7 ng/
mls 5: 104.2 ng/
mls 6: 107.3 ng/
Sequencing primers:
pSB416 GPD
M13 forward (-20) [46]
M13 reverse [47b]
pSBIC3
VF2 [125]
VR [126]
7/28/16
(HC)
Ran Spinpreps with QIAgen kit on cultures
(XX) Goal:
Miniprep culture using old QIAgene kit
Regrow culture
Digest and run a gel to test whether pSB416 GPD mls-yeGFP is successfully inserted?
Major steps:
Regrow culture: Add 4.5 mL LB-Amp broth into old culture
Digest and gel:
5 DNA
1 EcoRI-HF
1 PstI-HF
1 CutSmart Buffer
2 sterile water
Gel Order: L, 4A1, 4B1, 5B1, 6A1, 6B1, L
Gel results:
All minipreps worked and insert of mls-yeGFP into yeast vector pSB415 GPD was executed successfully!
Other activities:
Nanodrop pSB416 GPD mls-yeGFP minipreps:
4B1 = 410.4 ng/
4A1 = 283.1 ng/
5B1 = 175.7 ng/
6A1 = 413.4 ng/
6B1 = 168.2 ng/
Pick 4B1, 5B1, 6B1 for yeast transformation
DNA used:
4B1 = 2
5B1 = 3
6B1 = 3
Plate on CSM-URA:
Spread 50 on plate
Spread the rest of it on plate
7/29/16
(HC) Ran a digest
5 μL of DNA from A1
1 μL of XbaI
1 μL of XhoI
1 μL of Cutsmart buffer
2 μL of Sterile Water
Ran Gel extration from monach gel extraction kit
Ran transformations with
2μL of DNA
1μL of Fragment 16-1
10μL of Builder Master Mix
7μL of Sterile Water
(JM/CH)
Today we ran a gel extraction so that our plasmid was the only DNA in the transformation step of our E. coli
Mass of first tube: 0.990g
Mass of second tube: 0.986g
Mass of total 1: 1.225g
Mass of total 2: 1.189g
Mass of gel 1: 0.236g
Mass of gel 2: 0.203g
For gel extraction we followed the NEB protocol provided in the kit.
7/30/16
(HC) Inoculate Cultures of LB-Amp
7/31/16
(JM/CH)
Today we came in and ran a transformation of our new plasmid into competent E. coli cells.
We ran the protocol and then plated on LB Amp plates
8/1/16
(HC)Ran Spinprep with QIAgen kit.
Ran a digest with
2 μL of DNA
1 μL of PSTI
1 μL of Cutsmart buffer
6 μL of Sterile Water
2 μL of DNA
1 μL of XhoI
1 μL of Cutsmart buffer
6 μL of Sterile Water
Ran gel
1.)Ladder, 2.) 1 uncut, 3.) 1PstI, 4.) 1 XhoI, 5.) 2 uncut, 6.) 2 PstI, 7.) 2 XhoI, 8.)P413-PstI site uncut, 9.) P413-PstI site PstI, 10.)P413-PstI site XhoI
(JM)
We came in today and the plates had not grown up enough so today in lab I decided to do grunt work which included autoclaving tips and micro centrifuge tubes.
(XX)Made master plate with:
Parent strain with pSB416 empty plasmid
KO
Mls-yeGFP
8/2/16
(HC)Made Yeast competent cells.
(JM/CH)
Today our cells have grown enough in both of the plates 2-2 from tube 1 and tube 2.
We picked 2 colonies from each plate to make 4 cultures that will now grow overnight.
Looked into using chromophores to verify expression. Transformed the chromophore provided to us in the 2016 distribution kit (19E) into E. Coli and plated on LB Amp. to grow overnight
8/3/16
(JM/CH)
Yeah…… pSB1C3 is chloro resistant, not Amp. Re-plated the transformants onto LB Chloro plates and incubated overnight.
Used the QIAgen kit to prepare spinpreps of the transformants and ran digests with
1 μL of DNA
1 μL of PstI
1 μL of Cutsmart buffer
7 μL of Sterile Water
Uncut BE ligated products and one spinprep of 416CYC1 that was not BE ligsated wads cut with PstI were used as controls.


