HomePage • PROJECT • Composite Part
Composite Part
Favorite Composite Part
Split-HRP enzyme based PPI Detection device
Since protein-protein interactions (PPIs) have been reported to play important roles in signal transduction and gene expression, methods for monitoring PPIs in cells have been developed rapidly for years. In our registered and submitted parts, we provide two split-GFP fluorescence reporter systems, split-HRP enzymatic-based reporter systems and split Luciferase chemiluminescence-based reporter system.
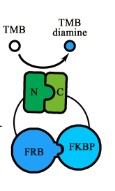
Our favorite composite part BBa_K1997011 is an important member of the collection PPI tool kit, which is constructed with the basic coding part (BBa_K1997005), lac promoter+RBS (BBa_K1789012), and double terminator (BBa_B0015) (Figure 1).
Figure 1. Schematic representation of the expressed proteins.
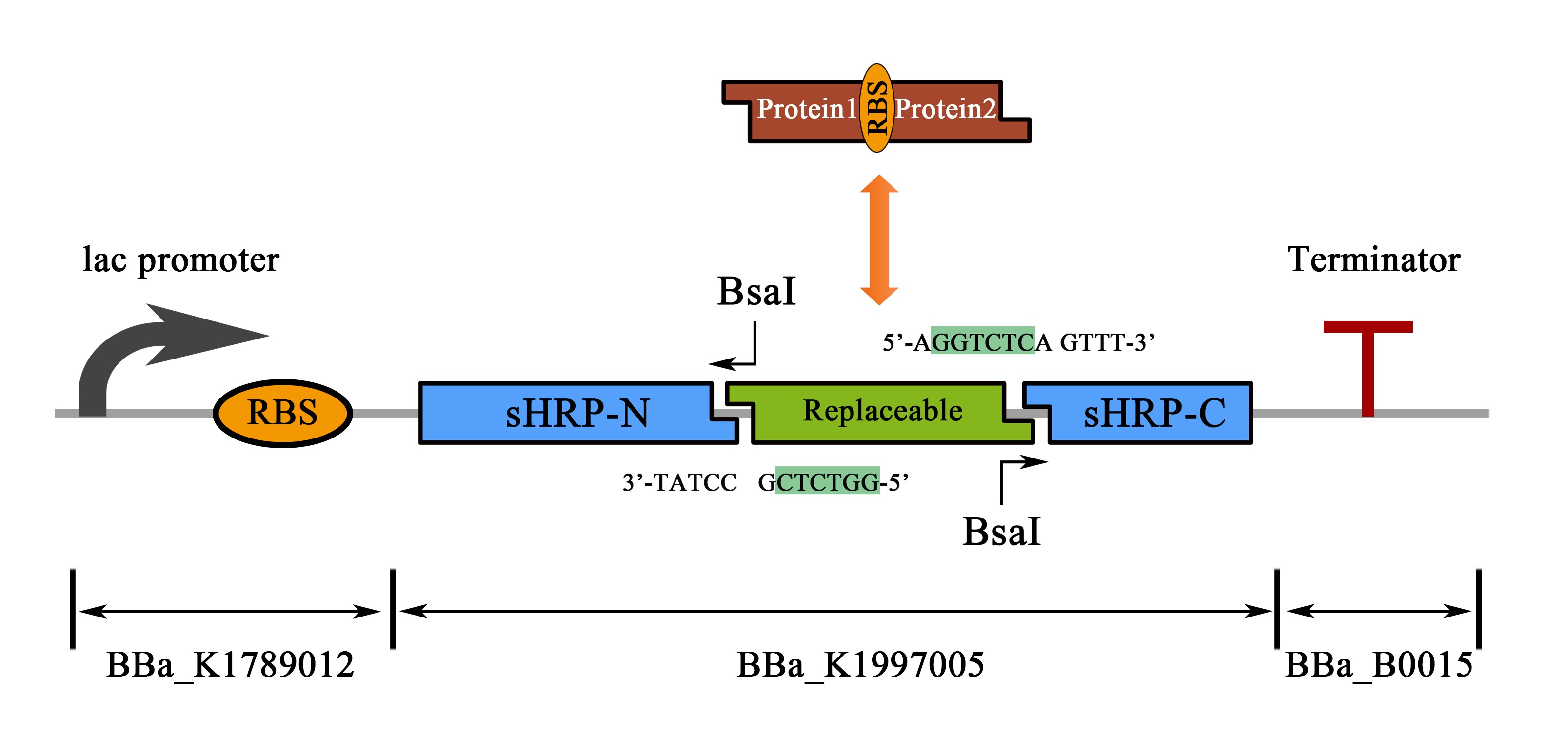
Actually, this composite part is a protein-protein interaction prototype of the “HRP-N-Zif269-HRP-C” part (BBa_K1997012). BBa_K1997012 contains a novel SUBSTITUTION SYSTEM for the construction of a protein-protein interaction reporter for any pair of proteins, so does this composite part. For instance, the replaceable FRB-RBS-FKBP segment in this composite part or the Zif268 segment in BBa_K1997012 can be replaced by other Protein1-RBS-Protein2 fragment following the procedures of Bsa I enzyme digestion and Golden-Gate Assembly (Figure 2). Thus, whether the protein1 and 2 are interaction proteins could be determined by output signal of HRP catalysis.
Figure 2. Schematic representation of the part design and the substitution system.
TO PROOF THE FUNTION of this composite part, we used Rapamycin to induce the interaction between FRB and FKBP. IPTG induced E.coli cells were then collected and lysed by high-pressure homogenizer. Once lysed and ultra-filtrated (to remove small molecules), 0.4nM of Rapamycin was added together with TMB substrate solution (with heme supplementation) into the cell lysate for the induction of protein-protein interaction and measurement of HRP activity. The plate was then incubated under 37°C for 5min before adding the Stop solution.
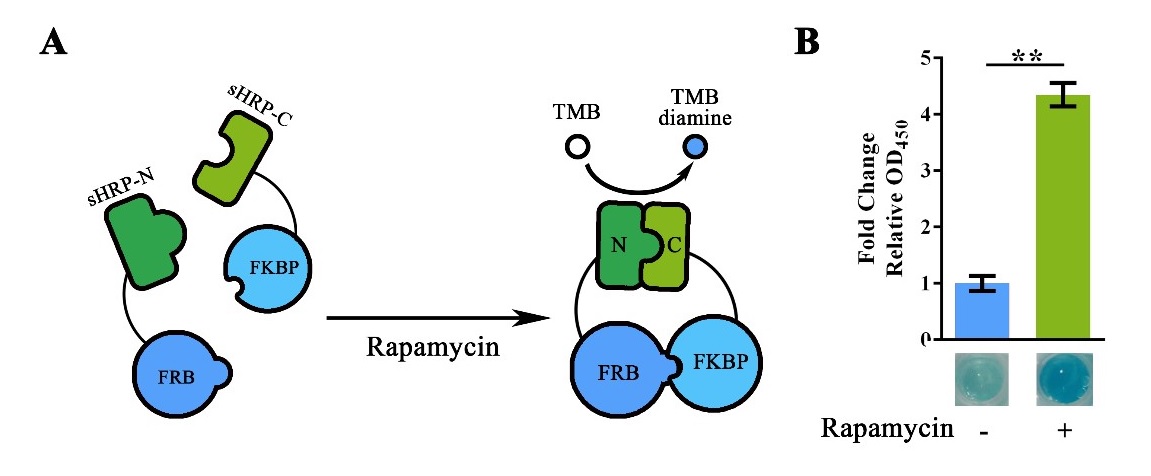
Value of OD450 obtained after adding the Stop Solution showed significant variation between the Rapamycin positive group and the Rapamycin negative group. Thus then validated the function of this part. (Figure 3).
Figure 3. Rapamycin-induced sHRP-N-FRB/sHRP-C-FKBP interaction.
(A) Schematic representation of the rapamycin induced protein-protein interaction. The adding of rapamycin would induce the interaction between FRB and FKBP, thus shortened the range between split-HRP fragments and reconstruct its structure for substrate processing. (B) HRP activity assay showing the fluorescent intensity with/without Rapamycin induction. For Fold change OD450, OD450of the group without Rapamycin induction was set arbitrarily as 1.0, and the levels of the other groups were adjusted correspondingly. The concentration of Rapamycin used in the experiment was 40nM. This experiment was run in three parallel reactions, and the data represent results obtained from at least three independent experiments. **p<0.01.
Collectively, this is an elaborate part for an optimal split-HRP reporting system, which was easy to be edit as a functional protein-protein interaction system.
Our Composite Parts
| NAME | TYPE | DISCRIPTION | LENGTH | SUBMITTED |
| BBa_K1997011 | Composite | P+R->sHRP-N->FRB->RBS->FKBP->sHRP-C->Ter | 2019 | yes |
| BBa_K1997012 | Composite | P+R->sHRP-N->Zif268-> sHRP-C->TER | 1638 | yes |
| BBa_K1997013 | Composite | P+R->sHRP-N->Zif-N->RBS->Zif-C->sHRP-C->TER | 1674 | yes |
| BBa_K1997015 | Composite | P+R->sGFP-N->Zif268->sGFP-C->TER | 1425 | yes |
| BBa_K1997016 | Composite | P+R->sGFP-N->Zif-N->RBS->Zif-C->sGFP-C->TER | 1461 | yes |
| BBa_K1997017 | Composite | P+R->sGFP-N->FRB->RBS->FKBP ->sGFP-C->TER | 1803 | yes |
| BBa_K1997018 | Composite | P+R->GFP10->Zif268->GFP11->RBS->GFP1-9->TER | 1485 | yes |
| BBa_K1997019 | Composite | P+R->GFP10->Zif-N->RBS->Zif-C->GFP11->RBS->GFP1-9->TER | 1521 | yes |
| BBa_K1997020 | Composite | P+R->GFP10->FRB->RBS->FKBP ->GFP11->RBS->GFP1-9-> TER | 1863 | yes |
| BBa_K1997021 | Composite | P+R->sLuc-N->Zif268->sLuc-C->TER | 2415 | yes |
| BBa_K19970022 | Composite | P+R->sLuc-N->Zif-N->RBS->Zif-C->sLuc-C->TER | 2451 | yes |
| BBa_K19970023 | Composite | P+R->sLuc-N->FRB->RBS->FKBP->sLuc-C->TER | 2793 | yes |
| BBa_K19970024 | Composite | P+R->sHRP-N->dCas9->Ter | 5180 | yes |
| BBa_K19970025 | Composite | P+R->sHRP-C->dCas9->Ter | 4823 | yes |