(→Introduction) |
(→Introduction) |
||
| Line 70: | Line 70: | ||
| − | = | + | =The role of herpes simplex virus thymidine kinase= |
Thymidine kinase plays an important role in pyrimidine salvage pathway. It catalyzes the monophosphorylation of thymidine to deoxythymidine monophosphate (dTMP) <ref>Kokoris, M. S., & Black, M. E. (2002). Characterization of herpes simplex virus type 1 thymidine kinase mutants engineered for improved ganciclovir or acyclovir activity. Protein science, 11(9), 2267-2272.</ref>. Human TK is incapable of phosphorylating guanosine analogues as ganciclovir, acyclovir, buciclovir, penciclovir and famciclovir. Since herpes simplex virus TK is able to phosphorylate guanosine analogues, it has been widely tested as a successful suicide gene in combination with ganciclovir, especially for the treatment of cancers and various virus infections, but also as a kill-switch in various iGEM-projects <ref> Moolten, F. L. (1986). Tumor chemosensitivity conferred by inserted herpes thymidine kinase genes: paradigm for a prospective cancer control strategy. Cancer research, 46(10), 5276-5281. </ref>. <br> | Thymidine kinase plays an important role in pyrimidine salvage pathway. It catalyzes the monophosphorylation of thymidine to deoxythymidine monophosphate (dTMP) <ref>Kokoris, M. S., & Black, M. E. (2002). Characterization of herpes simplex virus type 1 thymidine kinase mutants engineered for improved ganciclovir or acyclovir activity. Protein science, 11(9), 2267-2272.</ref>. Human TK is incapable of phosphorylating guanosine analogues as ganciclovir, acyclovir, buciclovir, penciclovir and famciclovir. Since herpes simplex virus TK is able to phosphorylate guanosine analogues, it has been widely tested as a successful suicide gene in combination with ganciclovir, especially for the treatment of cancers and various virus infections, but also as a kill-switch in various iGEM-projects <ref> Moolten, F. L. (1986). Tumor chemosensitivity conferred by inserted herpes thymidine kinase genes: paradigm for a prospective cancer control strategy. Cancer research, 46(10), 5276-5281. </ref>. <br> | ||
Revision as of 13:54, 18 October 2016
Safety
Introduction
Biosafety is defined as "the discipline addressing the safe handling and containment of infectious microorganisms and hazardous biological materials"[1] According to the U.S. Department of Health and Human Services and the CDC.
For our team the security and safety measures of our project have had the highest priority. Since there are many inherent risks of our work in the iGEM laboratory, we have decided to thoroughly present the dangers that we have analyzed while working at it. Firstly it is relevant to mention that we used non-pathogenic E. coli strains like BL21 and Xl1-blue for the cloning part of the project, which are typical strains used at the labs. Nevertheless we have had extreme caution to prevent any spreading of used living organisms, since this were S1 genetically modified organisms.
The field of synthetic biology provides us with the possibility to work with living organisms and modify them. There are many safety measures that are very important to consider while investigating and working at the laboratory for this purpose. Since the base of synthetic biology is the work with genetically modified organisms (GMO), it is necessary to look out for risk factors like spreading of the organism or infection from the investigators. It is also important to highlight that molecular biology works at the laboratory involves the use of some harmful chemicals, which is another aspect of the safety to be considered.
Safety in the laboratory
The Laboratory in which the iGEM Project has been conducted, at the Chair of biological chemistry is classified as a laboratory with the biosafety level S1. According to the German law of the regulation of GM technology (Gesetz zur Regelung der Gentechnik) there are 4 levels of biosafety, namely from S1 to S4. In the Level S1 there is considered that no risk for human health or the environment is present.[2] In order to guarantee the integrity of all the team members and the environment, we took preventive steps prior to the beginning of our lab work. All team members that were going to be performing wet lab tasks were required to attend to a lecture about the risks and precautions while working with GMOs. It was conducted by Dr. Martin Schlapschy in charge of the safety of the laboratory. In this presentation information about the regulation of biology by governmental institutions in Germany and the obligations each laboratory has to fulfill e.g. having a good and ethical practice was also included. After this all team members was given an introduction to the laboratory, in order to get to know all important safety features as well as localisation of the proper lab equipment. Factors like disposal of chemicals, GMOs and handling of dangerous chemicals or sensitive cells (cell culture) were specifically emphasized and thoroughly explained.
It is important to underline some important safety measures that have to be taken while conducting molecular biology experiments:
- The use of Ethidium bromide, since it is considered to be a mutagenic chemical compound. The use of gloves, and safety googles was obligatory while using it. [3]
a-rna-purification- analysis/nucleic-acid-gel-electrophoresis/dna-stains/ etbr.html This measures also applied for compound like Hydrochloric acid and Ammonia, additionally this were to be handled with the protection of a fume hood, due to the volatility of some of them.
- The use of a special protective mask in order to protect the eyes from the UV Light while analyzing gels.
- The use of certain machines at the laboratory posed some risks as well. As an example the use of big centrifuges, which if mishandled could cause harm to the investigators.
Biosafety: No danger to scientists, the population or the environment
In the daily work with genetically modified microorganisms and cells it is always important to consider various aspects of biosafety. When planning and conducting experiments one has to evaluate all potential risks and dangerous posed to human health and the environment thereby and take all eventual necessary precautionary measures (precaution principle). Under this principle general contact with biological material for instance shall be reduced to a minimum. As our project relies on a variety of wet lab work all constructs and experiments were carefully checked ahead of execution. Our work in the laboratory was determined not to pose any kind of unusual risks neither to the experimenters nor the general public or the environment. The strict containment of the experiments within the lab was ensured at all times. All strains and lines organisms and cells used were specifically designed for lab applications and are therefore not able to survive outside laboratory conditions as no aspect of our project work overrides these restraints.
Biosecurity: No potential misuse or dual-use character in our project
In order to fully ensure that a project does not allow for any harm to be dealt it is also imperative for the scientists to assess possible harmful misuse of an invention. Relevant to such a analysis are whether the genetic construct can be used directly or indirectly to arm microorganisms. Constructs for example that allow for the production of any kind of toxins or dangerous chemicals as well as genetic code that allows for the integration or transmission of DNA.
As we have evaluated our project accordingly we were not able to find any aspect of our work which could be used or modified to serve exterior unwanted purposes. No part allows for the production of any kind of harmful substances nor the spread of the construct themselves.
The debate surrounding bioprinting
The topic of bioprinting, due to the fact of being relatively young, is still currently being discussed in international ethics and safety forums and in courts. This is why we consider it to be essential to briefly present and discuss the main concerns around bioprinting. It is important to mention that there is a broad spectrum of perspectives and disciplines, in which key questions about tissue printing can be analyzed.
The world is facing a lot of challenges nowadays with the constant improvement and advances in the field of biology. These being questions about the ethic of some projects and furthermore the regulation of the reproduction of the same experiments, in the case of its success. This is why it is very important to consider all these political, social and ethical issues bonded to the scope and effect of the project. It is worth mentioning how there is a big debate about organ transplantation already since it first became possible in 1945 when the first kidney was successfully transplanted (HRSA). [4] With bioprinting a lot of new questions around patentability, affordability towards the different social strata, clinical safety and social acceptance arise and are to be discussed and dealt with.
One of the biggest security concerns around the use of 3D printed organs for transplantation is the tort liability of physicians, added to the intellectual property debate of 3D produced organs. Considering the fact that the transplantation of these organs needs still years of research of becoming a reality, the governments and regulation organisms are facing a great challenge in regulating the use and manufacture of 3D printed organs. To illustrate this problem, as an example, the Food and Drug Administration (FDA) is in charge of regulating medical devices as artificial hearts. Meanwhile is Organ Donation regulated by another governmental agreements, like in the case of Illinois, the Anatomical gift act. [5]
Interview with member of national biosafety panel (ZKBS) Professor Dr. Alfons Gierl
This year we invited Professor Dr. Alfons Gierl, a german scientist in the field of genetics and a member of the [http://www.bvl.bund.de/DE/06_Gentechnik/02_Verbraucher/05_Institutionen_fuer_biologische_Sicherheit/02_ZKBS/gentechnik_zkbs_node.html national biosafety panel (ZKBS)] to discuss safety matters of our project but also talk about the potential and future of synthetic biology and his experience in the field of GMO's. In his opinion synthetic biology can be a promising field of research, if the main goal is application in the biomedical area since the demand in this area is very high and has the best chance to be accepted by the non-scientist community. In safety questions the [http://www.bvl.bund.de/DE/06_Gentechnik/02_Verbraucher/05_Institutionen_fuer_biologische_Sicherheit/02_ZKBS/gentechnik_zkbs_node.html ZKBS] plays an important role in germany as it, for example, paraphrases the law to evaluate what risk group a certain GMO belongs to. As he says the current laws in Germany cover most of the recent developments and he is certain that they will cover the developments in synthetic biology for the next couple of years as well. Since our project deals with the generations of GMO's that can be printed in 3D tissues and could possibly be implanted into a recipient we wanted to know what he thinks about the risk of GMO release. According to him the risk is rather small, since the constructs are integrated stabily and the GMO's do not have contact with cells of the germline. There were many more topics covered, that can not be mentioned here. We want to thank Professor Dr. Gierl again for his support and this highly interesting interview.
Interview with Dr. Nina Köhler, Bavarian Health and Food Safety Authority (LGL)
During our Synthetic Biology project we have held an interview with Dr. Nina Köhler from the Bavarian Health and Food Safety Authority (LGL). Dr. Köhler is working on a project monitoring the current national and international developments in synthetic biology and on developing strategies for their analytical surveillance. Topics of the interview were the current regulations of Synthetic Biology in the EU and in Germany. Dr. Köhler referred to the “Zentrale Kommission für die Biologische Sicherheit” (ZKBS) at the Federal Office of Consumer Protection and Food Safety (BVL). According to the ZKBS all current Synthetic Biology activities (apart from in vitro gene/genome synthesis and protocell research) are regulated by the German Genetic Engineering Act.
We want to thank Dr. Köhler for taking the time and talking to us about our project.
A practical contribution to Biosafety in iGEM projects: The kill-switch
Aim of the new designed Thymidine kinase / ganciclovir system
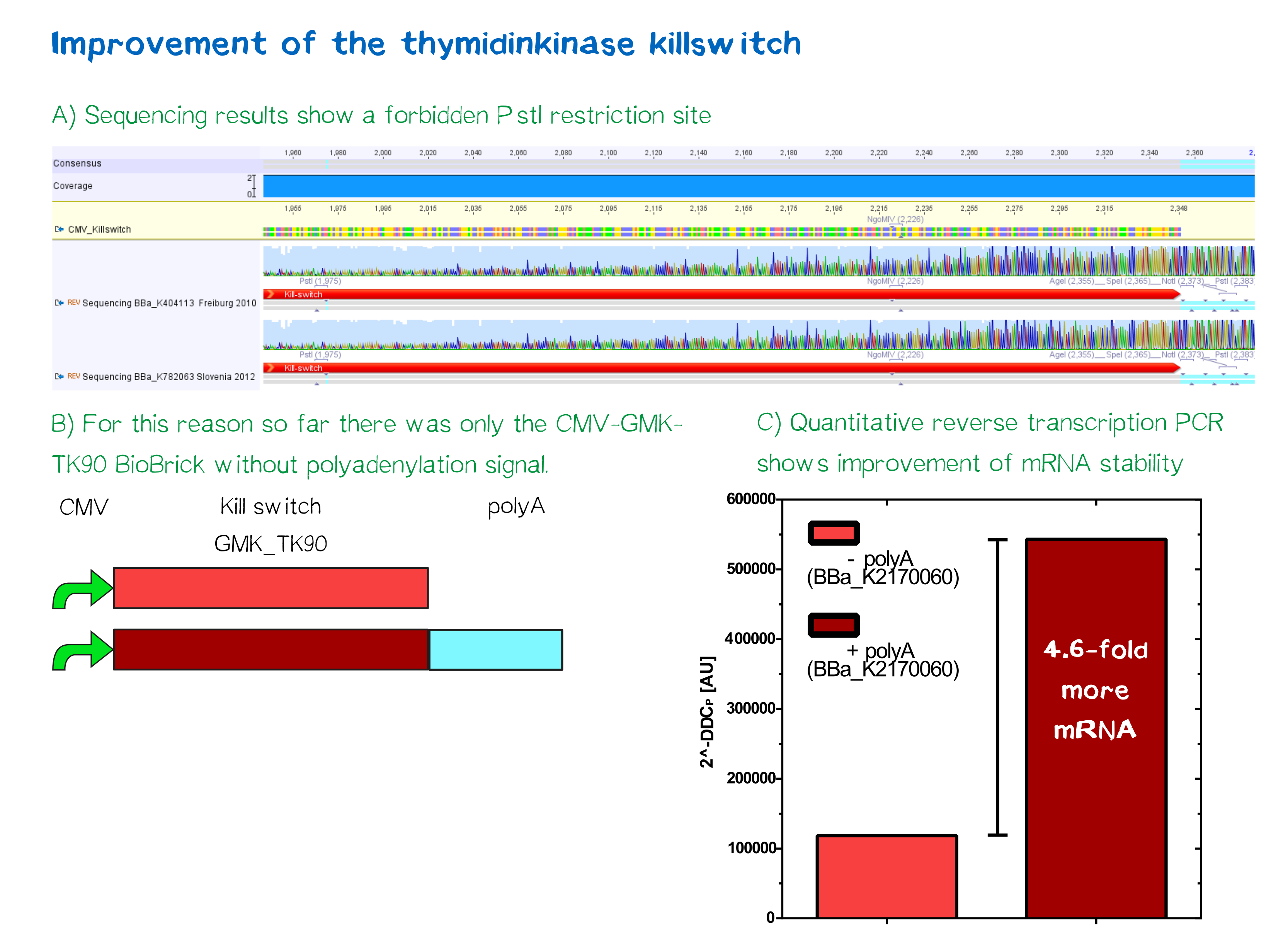
One key factor of guaranteeing the safety of our project and preventing in advance any dangers, is the implementation of a well-known kill-switch. In this case we worked in cooperation with the iGEM-team of Slovenia and Freiburg, who each provided us with their Biobrick (BBa_K404113 - HSV-thymidine kinase fused to mouse guanylate kinase, iGEM Freiburg 2010 and BBa_K782063 - CMV-promoter, iGEM Slovenia 2012). This specific kill switch is known as the Thymidine kinase / ganciclovir system. In order to enhance the current Biobrick, we have repaired a point mutation that led otherwise to a pstI restriction location. An additional Poly-A-tail should also stabilize the mRNA later on. For more details, see our experiment protocol further down.
The role of herpes simplex virus thymidine kinase
Thymidine kinase plays an important role in pyrimidine salvage pathway. It catalyzes the monophosphorylation of thymidine to deoxythymidine monophosphate (dTMP) [6]. Human TK is incapable of phosphorylating guanosine analogues as ganciclovir, acyclovir, buciclovir, penciclovir and famciclovir. Since herpes simplex virus TK is able to phosphorylate guanosine analogues, it has been widely tested as a successful suicide gene in combination with ganciclovir, especially for the treatment of cancers and various virus infections, but also as a kill-switch in various iGEM-projects [7].
Freiburg 2010 has first implemented the fusion protein of HSVTK since it conveys higher toxicity results. Plain HSVTK has only limited kinase activity on guanosine nucleosides. To compare GCV to thymidine, it is 500-fold more catalytic active in the case of thymidine than GCV. The specific mutant 30 of HSVTK attributes besides the fusion with mGMK to an even higher improvement in toxicity. For iGEM, it may be also of great interest to take the mouse GMK since it is soluble in E. coli in comparison to its human analogue [8].
After successful transfection of mGMK-TK30 fusion protein the following incubation with one of the mentioned guanosine analogues leads initially to monophosphorylation followed by di- and trimethylation by cellular kinases. The trimethylated state is obviously the active form of the prodrugs [9]. Triphosphorylated guanosine analogs like GCV can be incorporated into nascent DNA and lead to inhibition of DNA replication. This leads to cell death [10].
References
- ↑ U.S. Department of Health and Human Services (2009, December). Biosafety in Microbiological and Biomedical Laboratories. Retrieved October 6, 2016, from http://www.cdc.gov/biosafety/publications/bmbl5/BMBL.pdf
- ↑ Bundesministerium der Justiz und für Verbraucherschutz. Gesetz zur Regelung der Gentechnik (Gentechnikgesetz - GenTG)§ 7 Sicherheitsstufen, Sicherheitsmaßnahmen. Retrieved October 06, 2016, from http://www.gesetze-im-internet.de/gentg/__7.html
- ↑ Ethidium Bromide. Retrieved October 06, 2016, from https://www.thermofisher.com/de/de/home/life-science/dn
- ↑ Timeline of Historical Events Significant Milestones in Organ Donation and Transplantation. Retrieved September 03, 2016, from http://www.organdonor.gov/legislation/timeline.html
- ↑ Park, M. H. For a new heart, just click print. Retrieved September 10, 2016, from http://www.globalforumljd.org/cops/legal-and-ethical-aspects-human-organs-3d-bioprinting
- ↑ Kokoris, M. S., & Black, M. E. (2002). Characterization of herpes simplex virus type 1 thymidine kinase mutants engineered for improved ganciclovir or acyclovir activity. Protein science, 11(9), 2267-2272.
- ↑ Moolten, F. L. (1986). Tumor chemosensitivity conferred by inserted herpes thymidine kinase genes: paradigm for a prospective cancer control strategy. Cancer research, 46(10), 5276-5281.
- ↑ Ardiani, A., Sanchez-Bonilla, M., & Black, M. E. (2010). Fusion enzymes containing HSV-1 thymidine kinase mutants and guanylate kinase enhance prodrug sensitivity in vitro and in vivo. Cancer gene therapy, 17(2), 86-96.
- ↑ Miller, W. H., & Miller, R. L. (1980). Phosphorylation of acyclovir (acycloguanosine) monophosphate by GMP kinase. Journal of Biological Chemistry, 255(15), 7204-7207.
- ↑ Reardon, J. E. (1989). Herpes simplex virus type 1 and human DNA polymerase interactions with 2'-deoxyguanosine 5'-triphosphate analogues. Kinetics of incorporation into DNA and induction of inhibition. Journal of Biological Chemistry, 264(32), 19039-19044.