In the notebook, there are some simplifications we made in sample names:
| Orignal | Simplified |
| ADH1 promoter( pADH1) | ADH |
| GAL1 promoter (pGAL1) | GAL |
| CUP1 promoter (pCUP1) | CU |
| Nano-lantern cAMP 1.6 | NLc |
| human adrenergic receptor beta 2 (ADRB2) | hAR |
| yeast nucleotide binding alpha subunit (Gpa2) with 5 amino acids modification | Gpa2M |
| Yeast TRP section marker | TRP |
| Yeast HIS section marker | HIS |
| Yeast LEU section marker | LEU |
| 6 x His-tag | his |
Contents
20160624
- Designed primers for amplifying the ADH1 promoter from yeast genome .
- Our oder (Nano-lantern cAMP 1.6) from addgene arrived!
20160625
- pick up colonies of pcDNA3-Nanolantern.
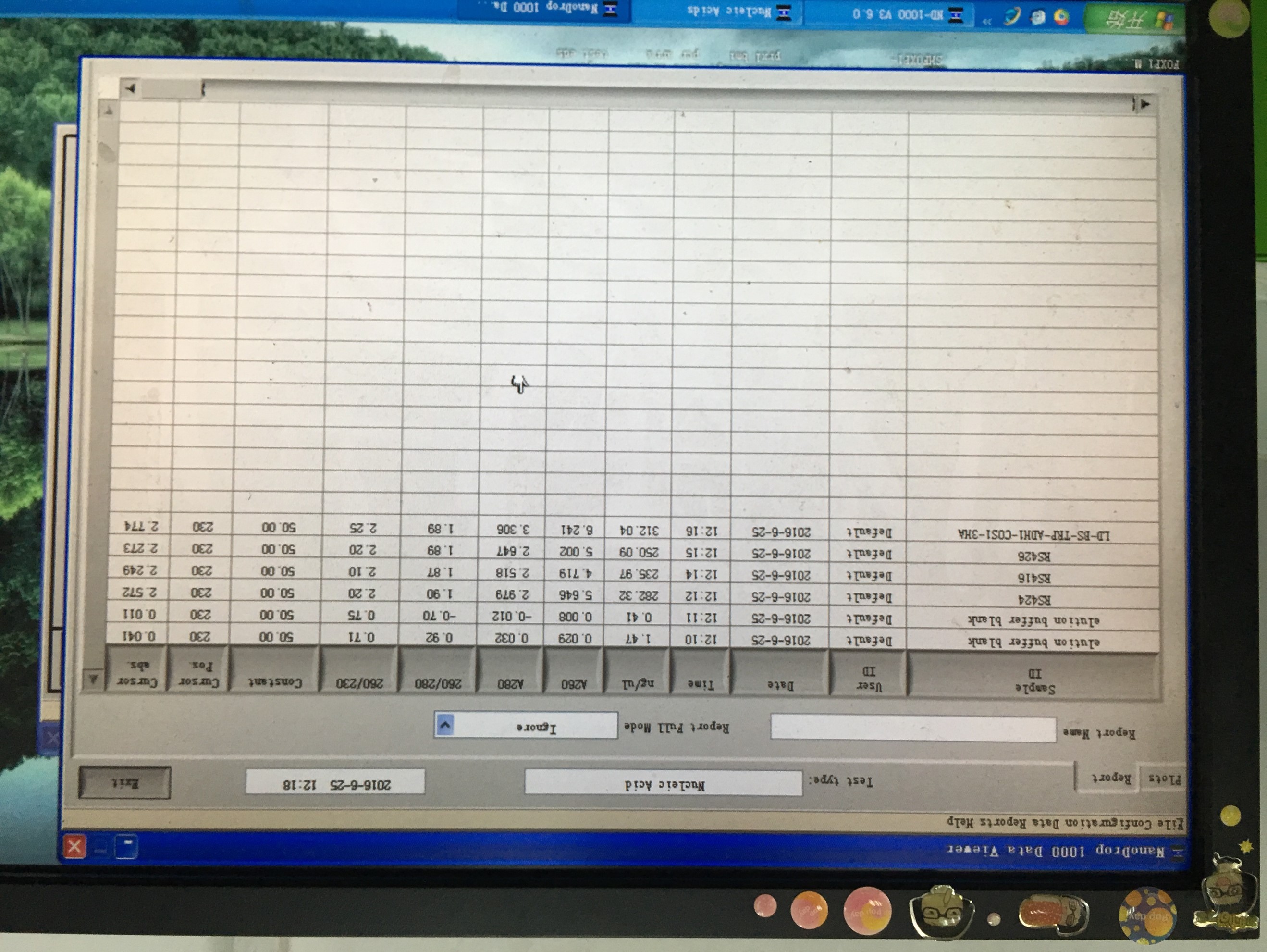
- Plasmid extraction of RS416、426、424、LD-BS-ADH1-COS1( plasmid backbones)
20160626
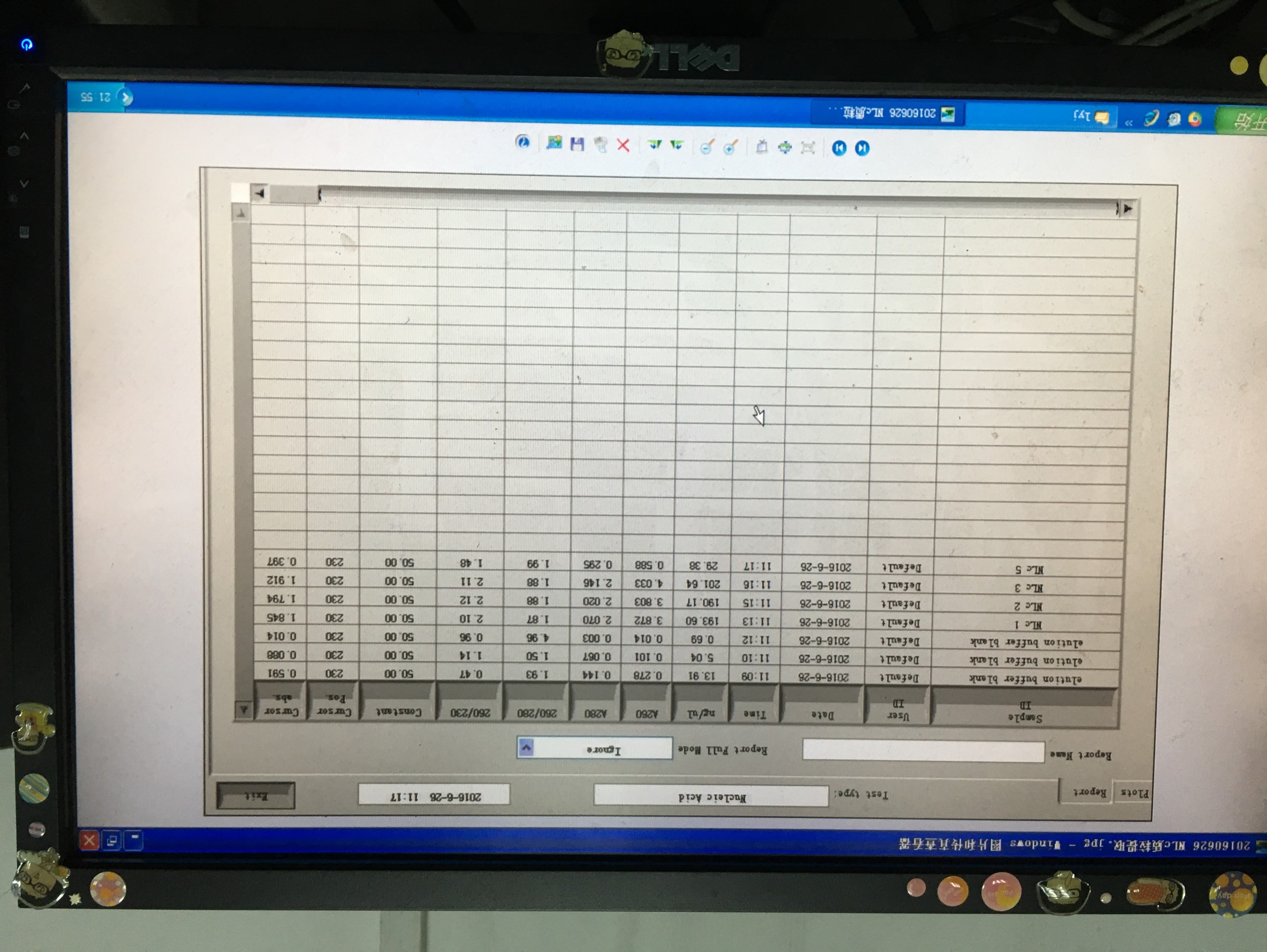
- Plasmid extraction of pcDNA3-Nanolantern
- ClaI-AvrII restriction digestion of LD-BS-ADH1-COS1
- PCR amplification of Nano-lantern cAMP 1.6 from pcDNA3-Nanolantern
20160629
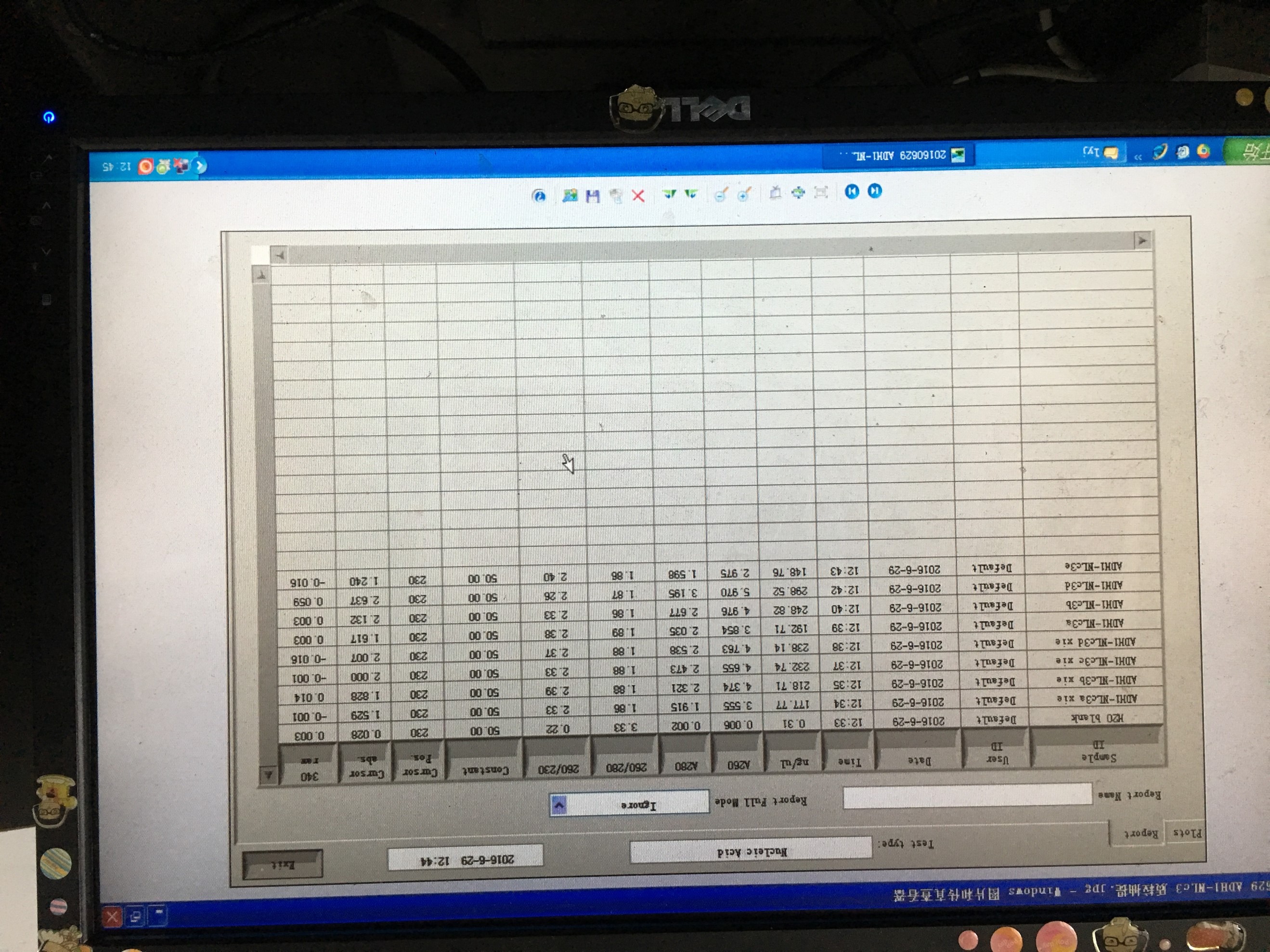
- Plasmid extraction of TRP-pADH1-NLc
- PCR amplify the GAL1 and CUP1 promoter from yeast genomic DNA and cloned them into plasmid backbones.
20160630
- Pick up colonies of GAL1 and CUP1 promoter with ADH1 terminator (TRP-CU/TRP-GAL).
20160701
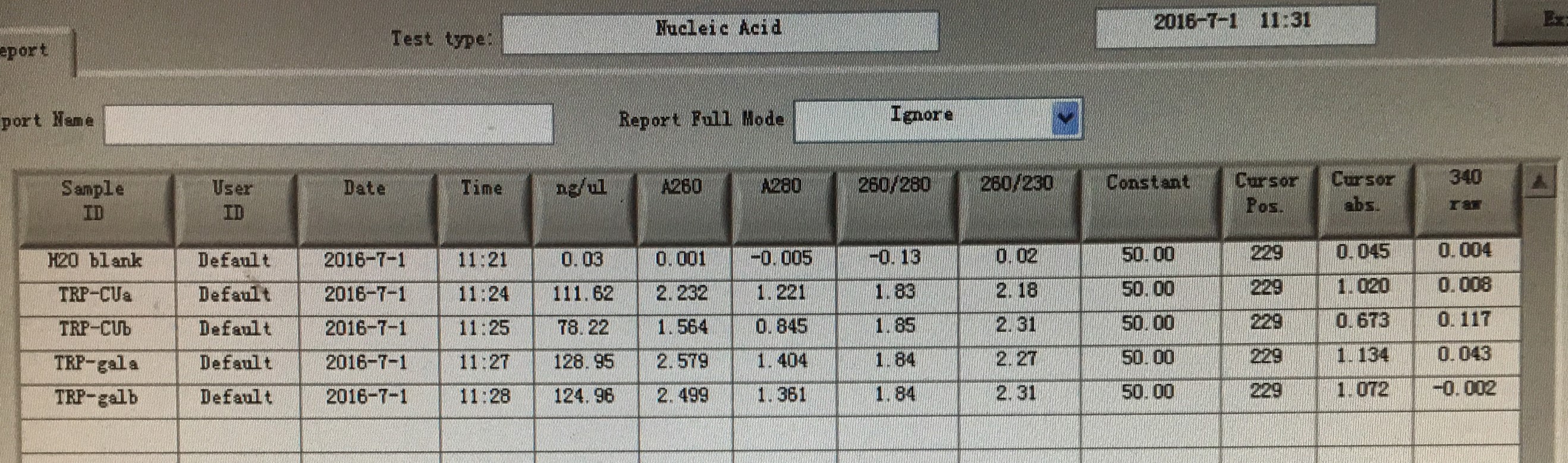
- Plasmid extraction of TRP-CU/TRP-GAL.-
20160706
- Got strain DJ03 and 9060 from professor Zhiping Xie’s lab.
- Ordered coelenterazine-h from YEASEN BIOTECHNOLOGY.
20160707
- Transformation of DJ03 and 9060 with construct TRP-ADH-Nlc.
| Volume | OD600 | Control/Experimetnal | Digested DNA | |
| DJ03 | 3ml | 0.9 | 1ml/2ml | 0μl/10μl |
| 9060 | 3ml | 0.8 | 1ml/2ml | 0μl/10μl |
20160709
- Seeking for the cds for human adrenergic receptor beta 2 (ADRB2)
20160710
- Making modification plans for heterologous expression of ADRB2 in yeast.
20160711~20160713
- Finishing the construct TRP-CU-NLc and TRP-CU-NLc
20160713
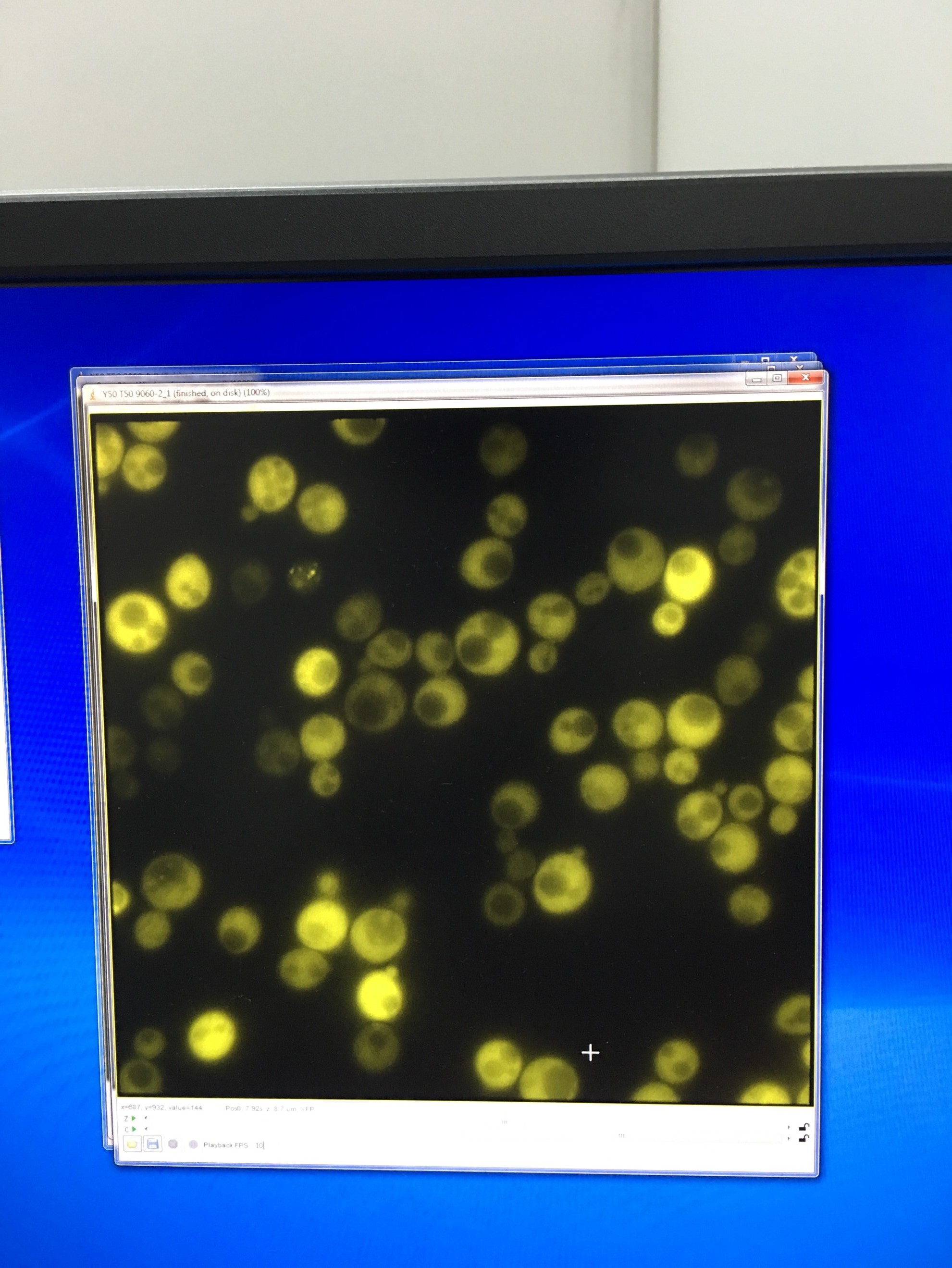
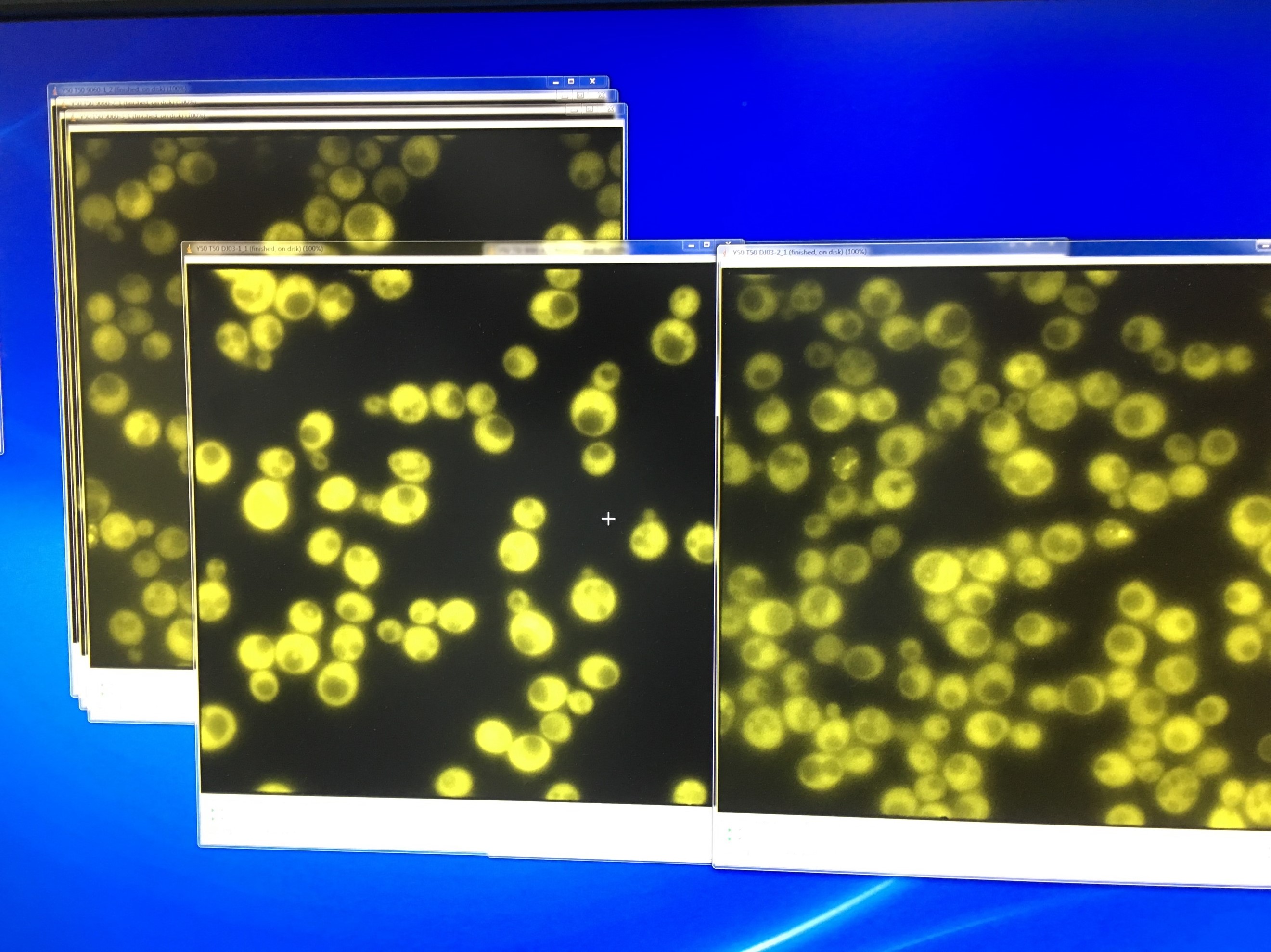
- Fluorescent microscopy of TRP-ADH1-NLc to detect the YFP signal
20160714
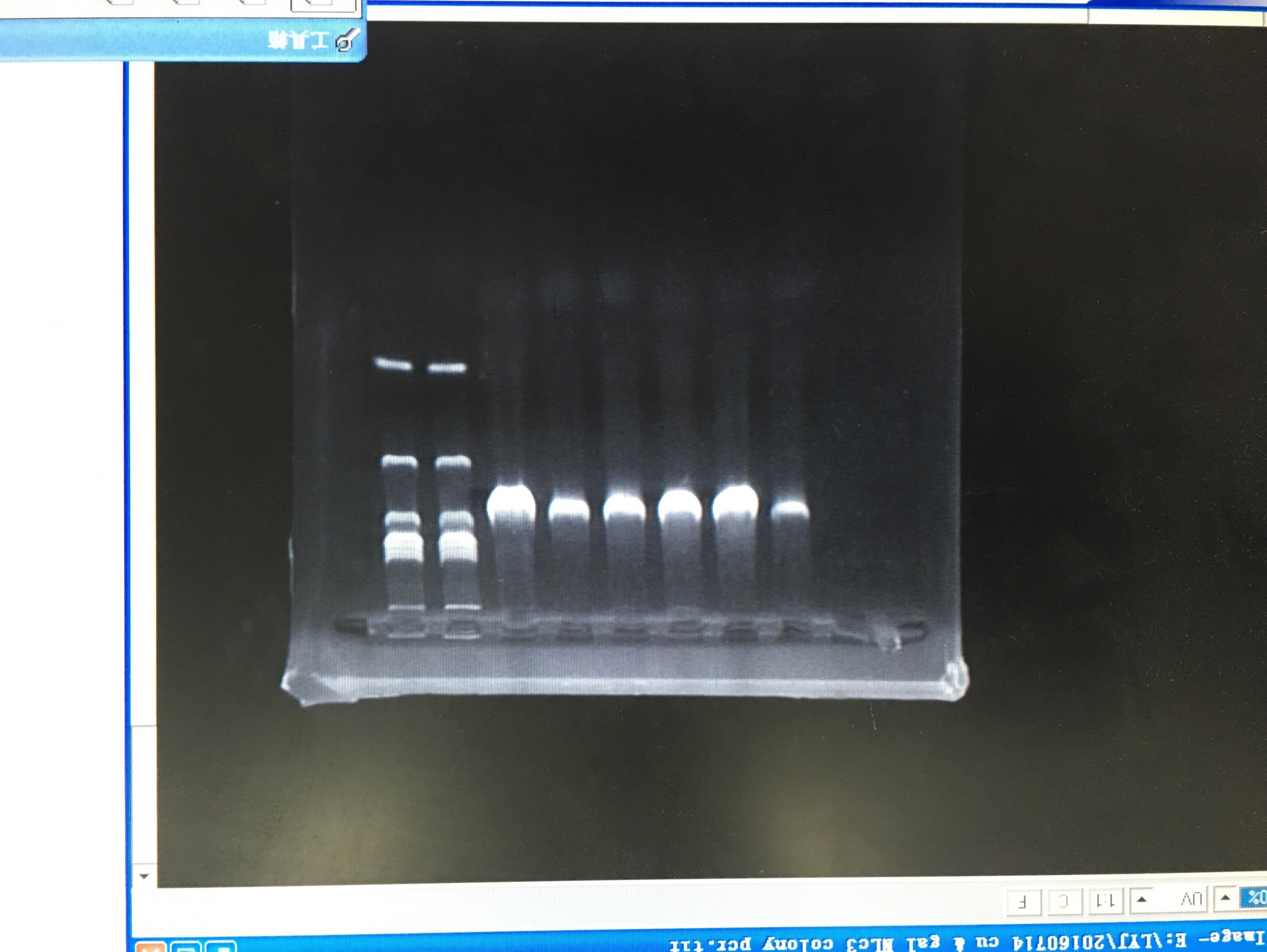
- Colony PCR of TRP-GAL-NLc3 abc/TRP-CU-NLc3 abc
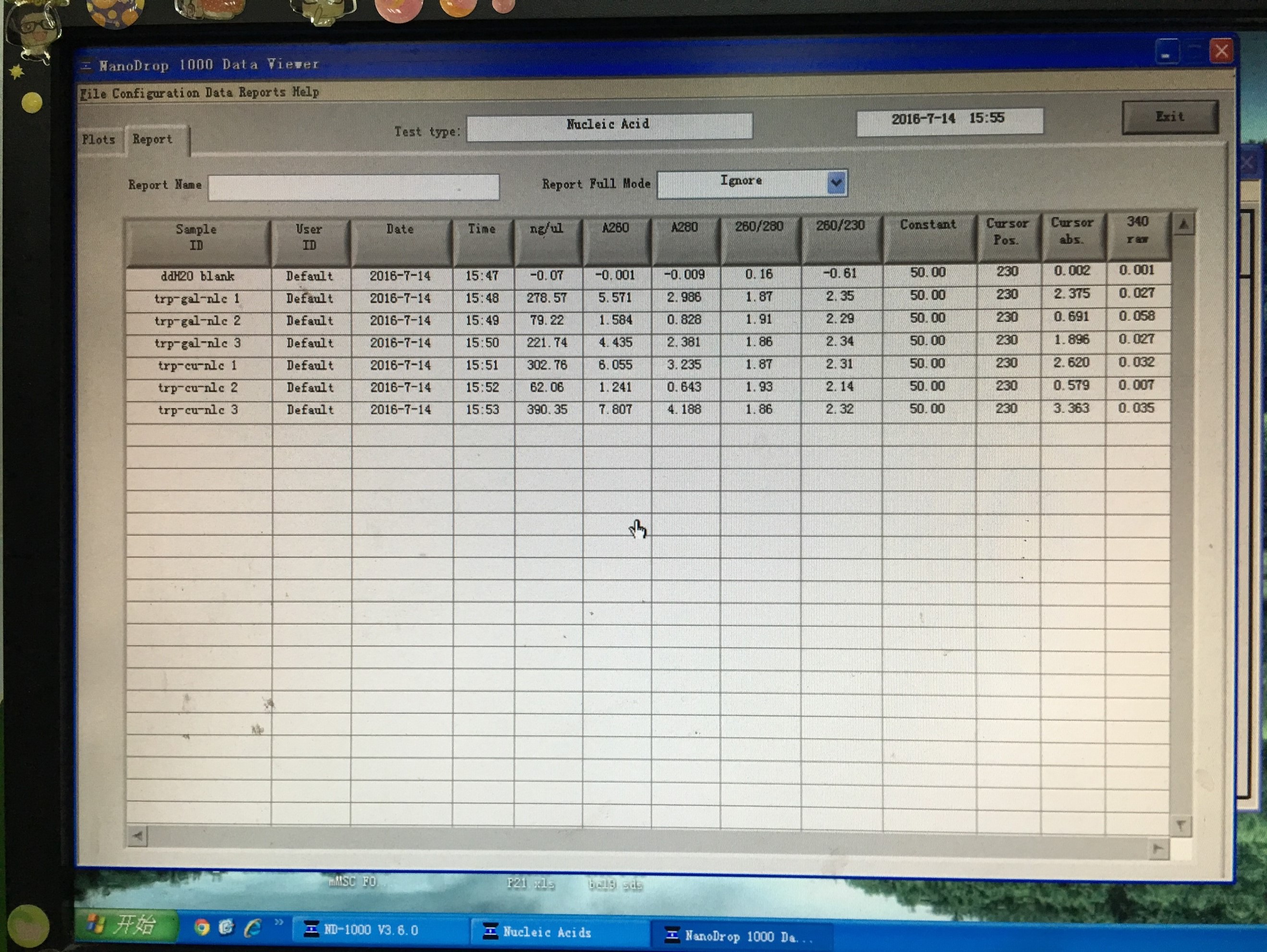
- Plasmid extraction of TRP-GAL-NLc3 abc/TRP-CU-NLc3 abc
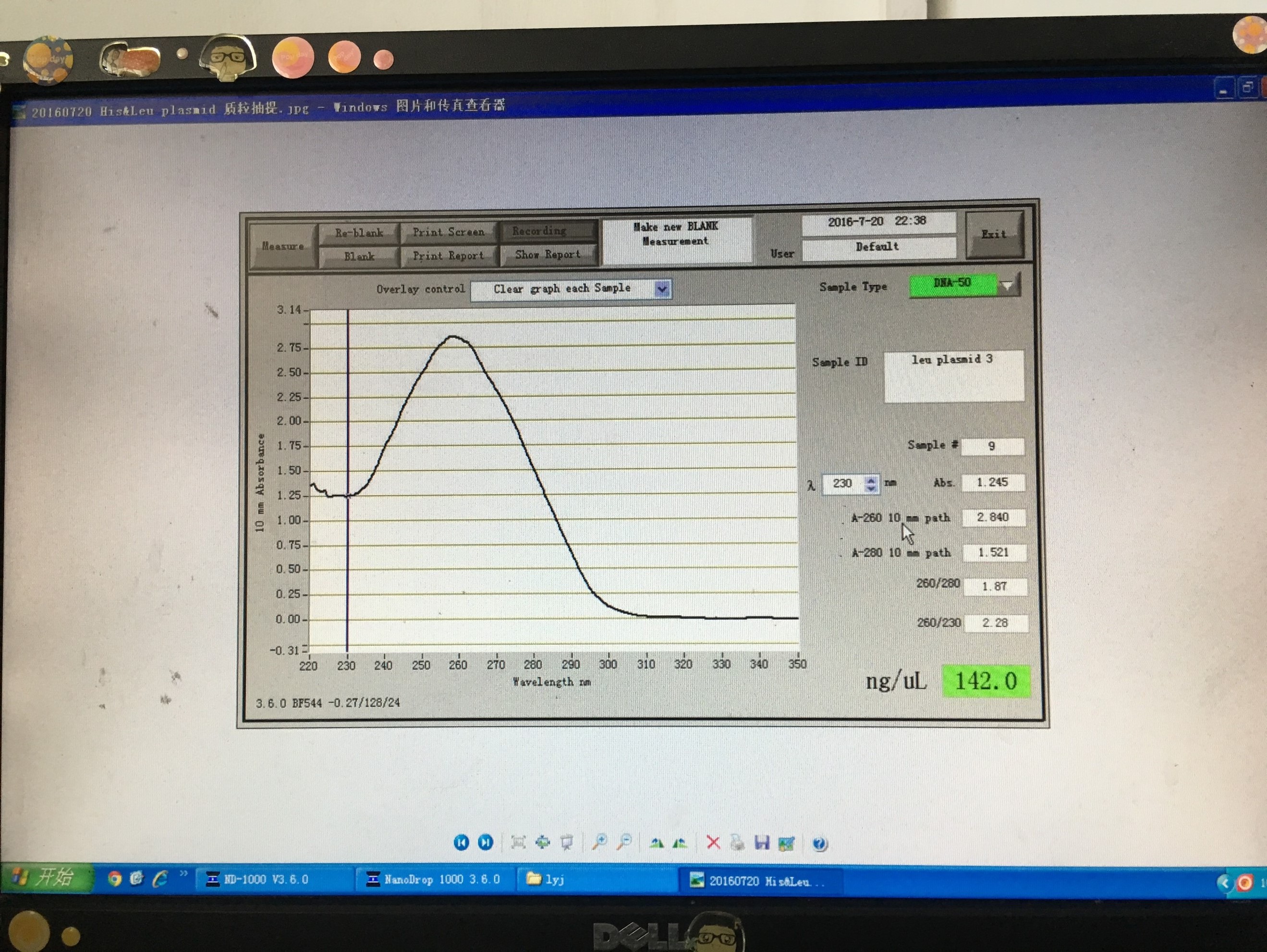
20160719~20160720
- Obtained plasmid backbone LD-BS-HIS3 and LD-BS-LEU2 from Zhiping Xie’s lab and amplified them to make larger amount.
20160723
- Tried to amplify the ADBR2 cds from genomic DNA of A549 cells
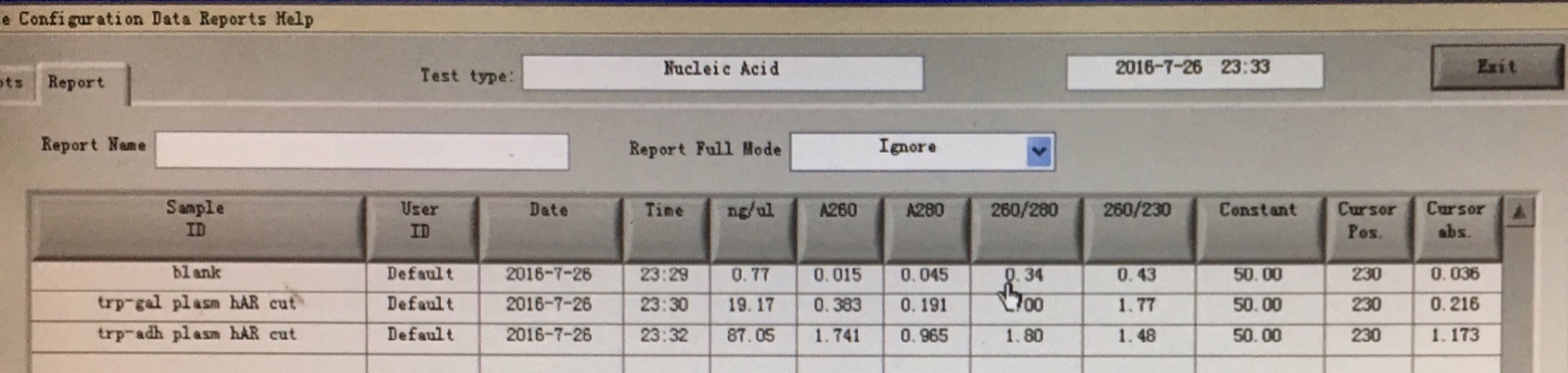
20160726
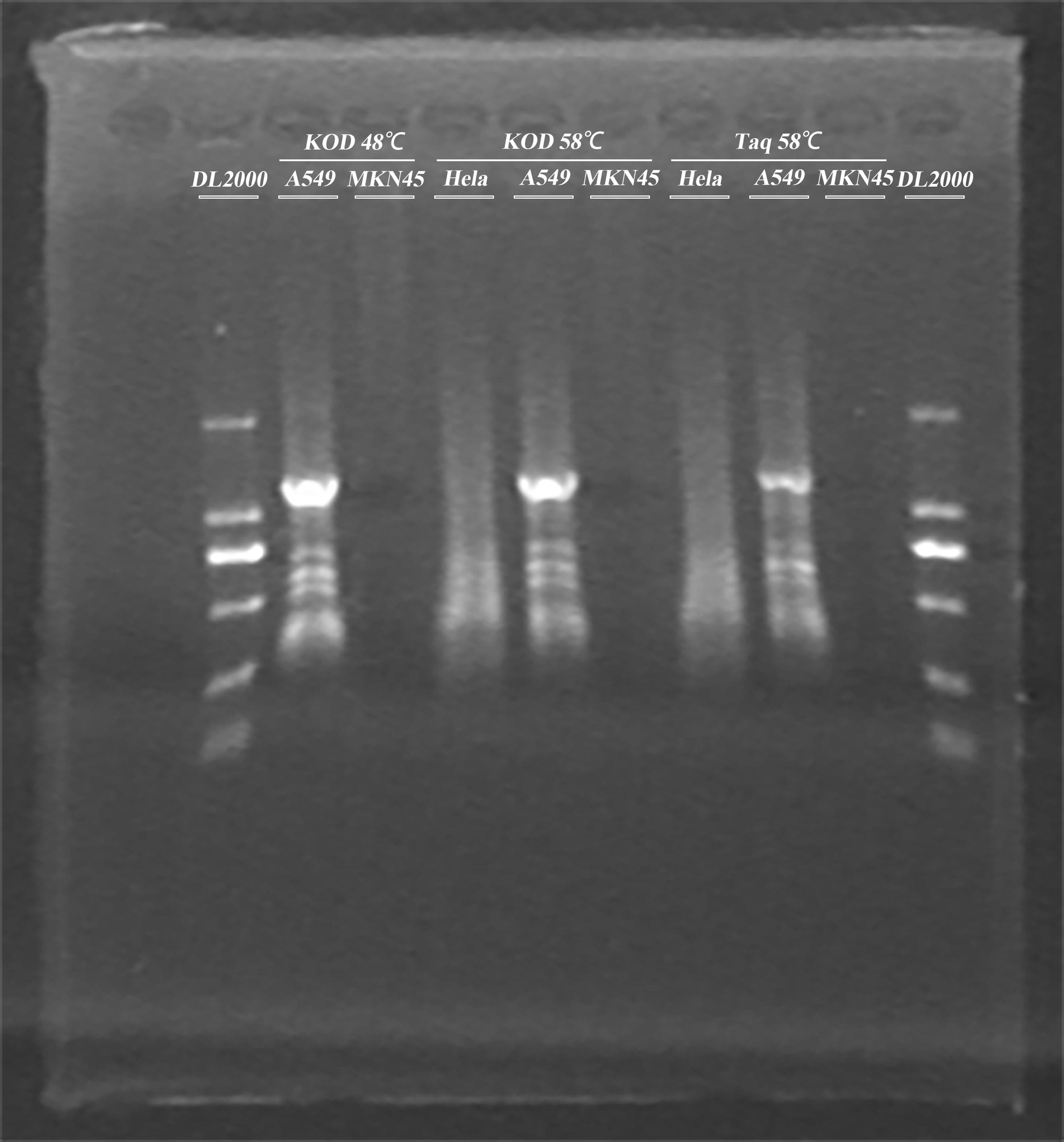
- Amplify the ADRB2 gene from genomic DNA of A549 cells using different melting temperature and different polymerase
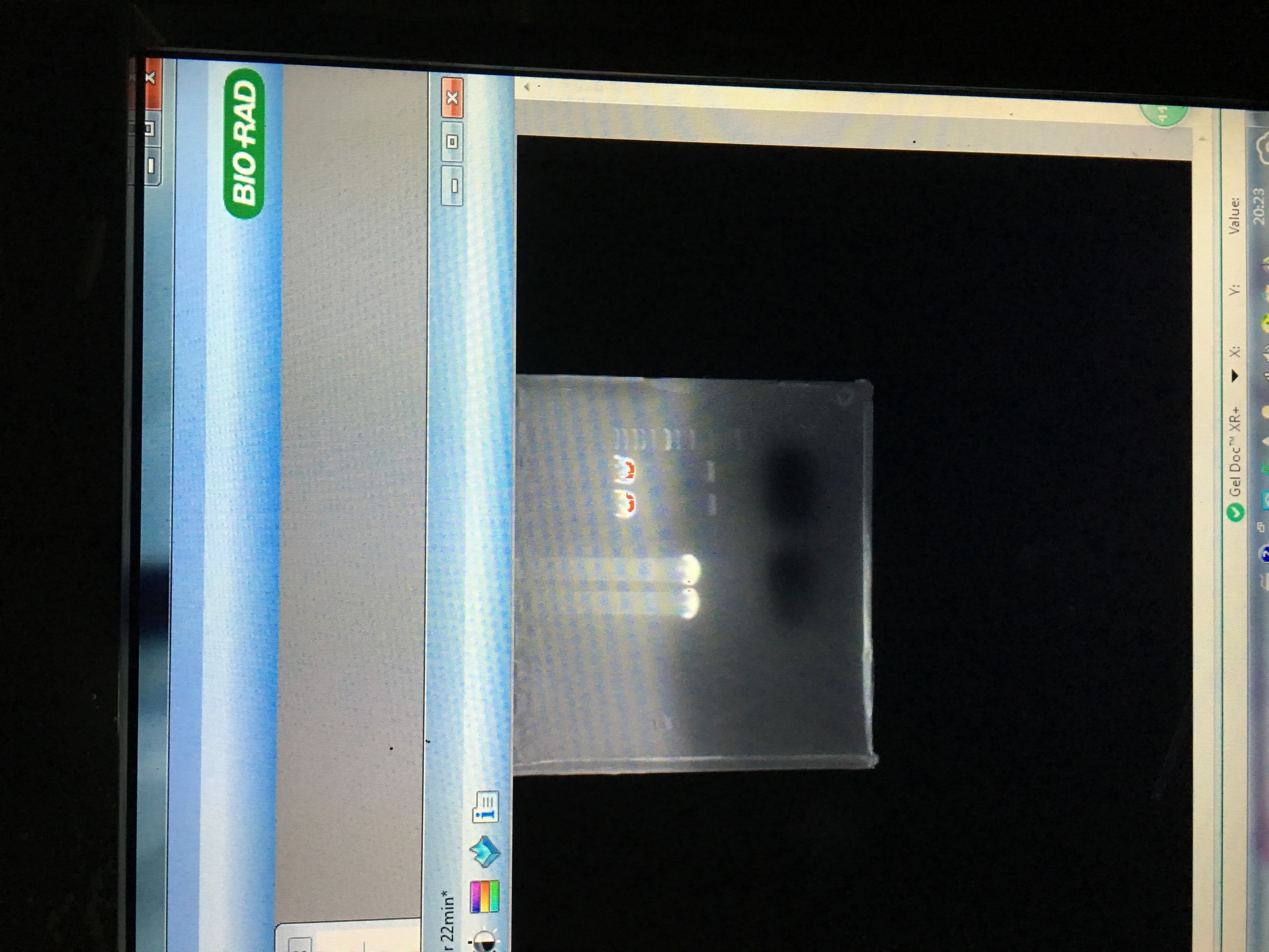
- Gel extortion of ADRB2 gene
- Enzyme digestion of LD-BS-TRP-pADH1-CDS1-3HA and LD-BS-TRP-GAL-3HA-DGA1-aid-6HA
- Amplify the ADRB2 gene from the fragment from gel extraction product
| Reagents | Volume |
| KOD plus neo | 1 μl |
| 10x Buffer for KOD -Plus- Neo | 5 μl |
| 2mM dNTPs | 5 μl |
| 25mM MgSO4 | 3 μl |
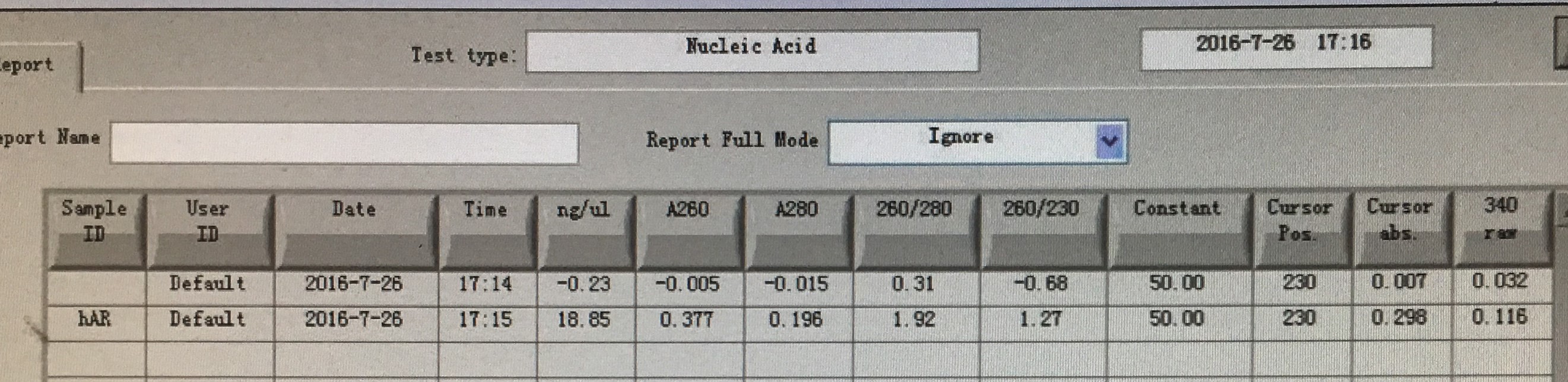
| hAR | 10 μl |
| 10 pmol/µl Primer | 1x2 μl |
| ddH2O | 24 μl |
Primer: hAR : hAR-R/hAR-F TRP-ADH-hAR : TRP-ADH-hAR-R/ TRP-ADH-hAR-F TRP-GAL-hAR : TRP-GAL-hAR-R/ TRP-GAL-hAR-F
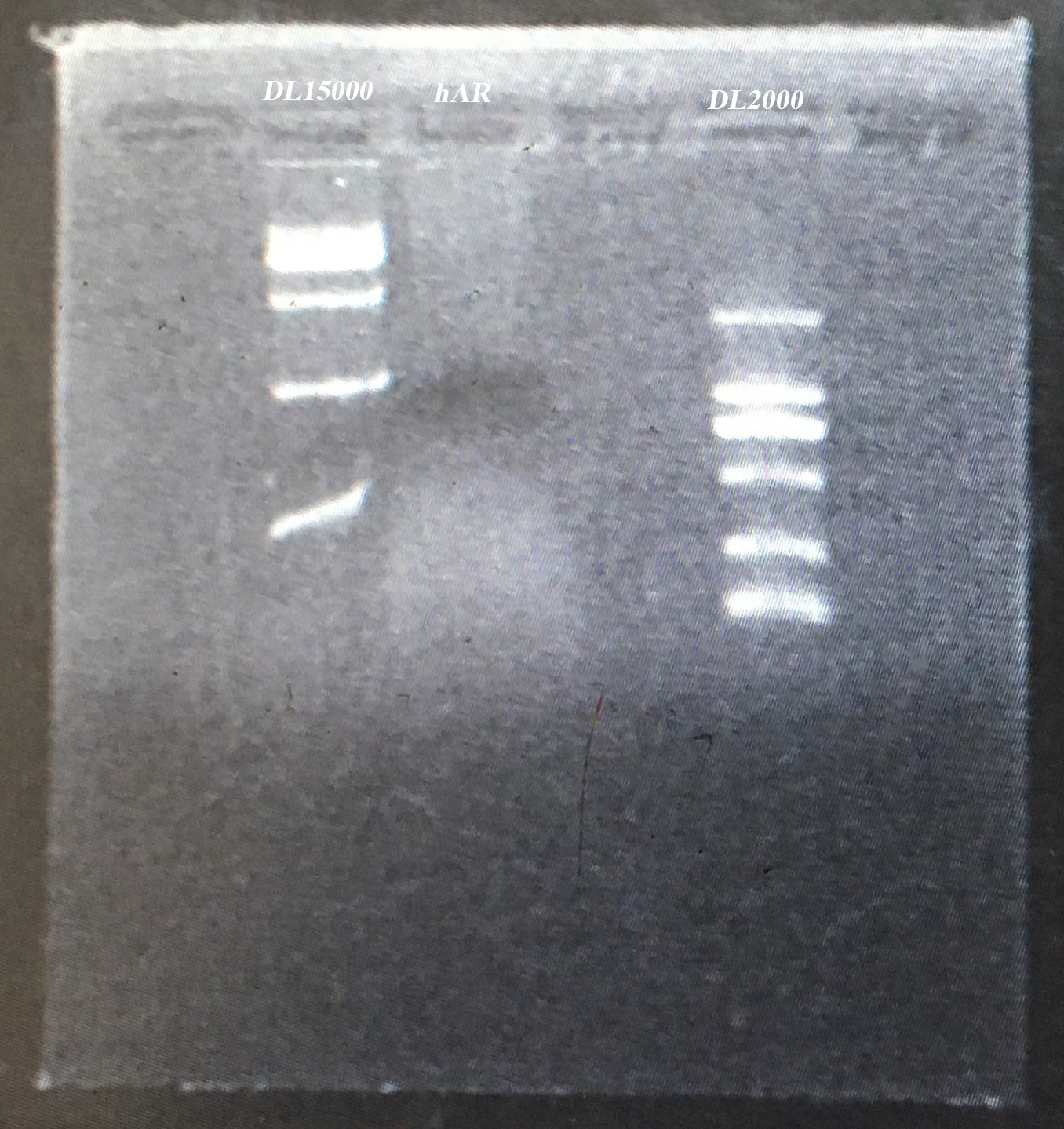
- Gel electrophoresis of the PCR product in 5 and endonuclease digested product in 4
- Gel extraction of endonuclease digested product
20160727
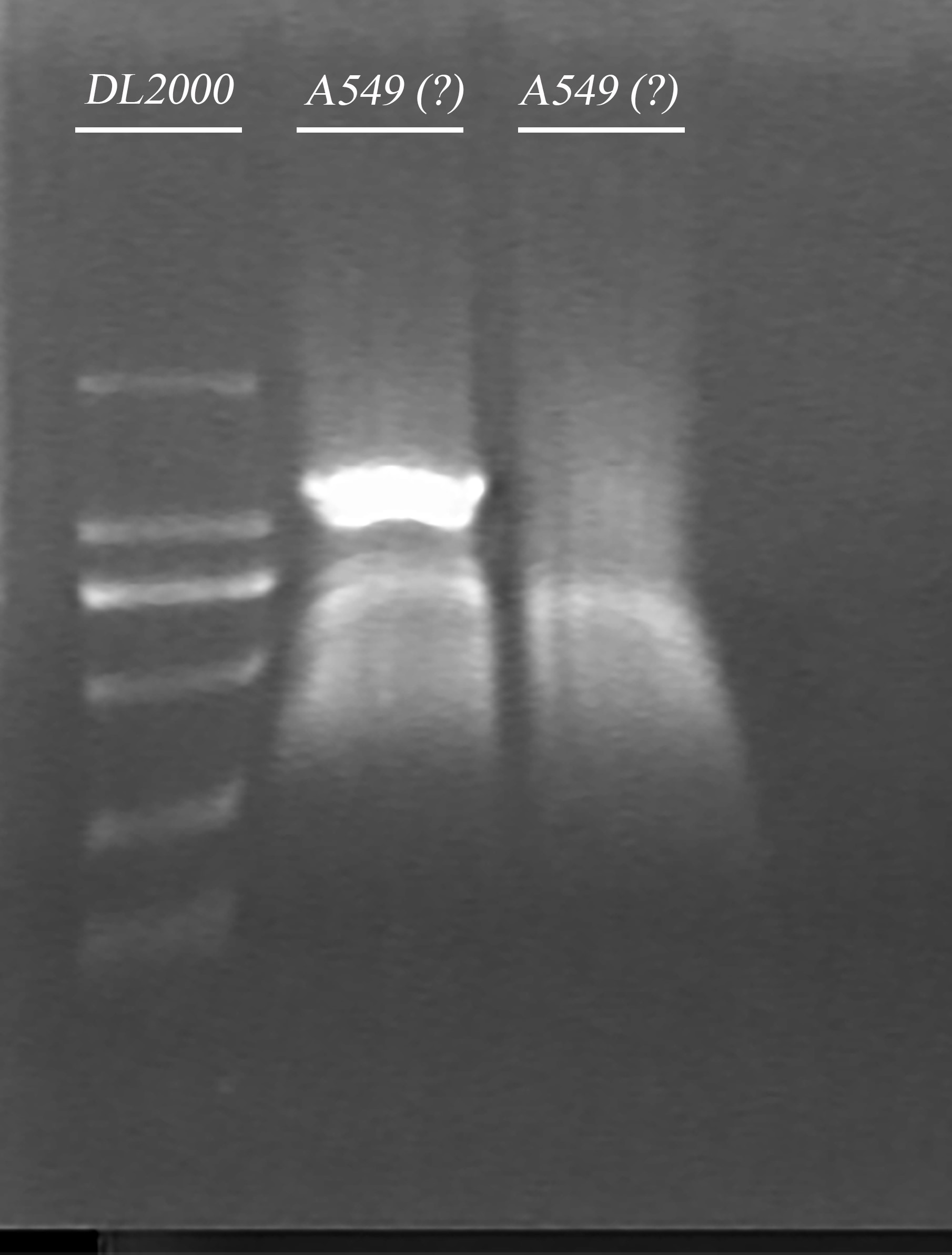
- PCR amplification of ADRB2 gene.
- PCR cleaning
- Gel electrophoresis of product in 3.
- Gel extraction
20160728
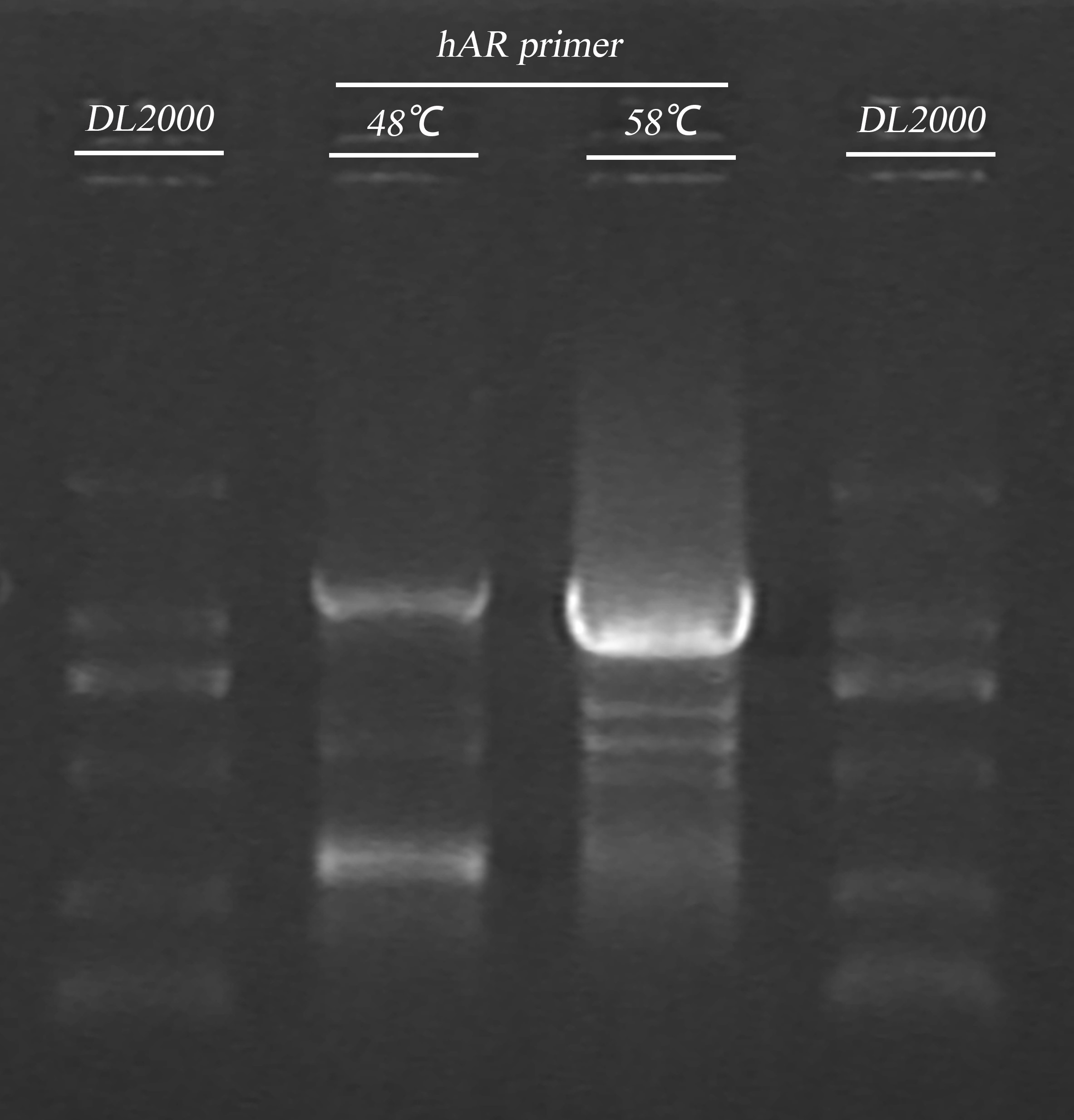
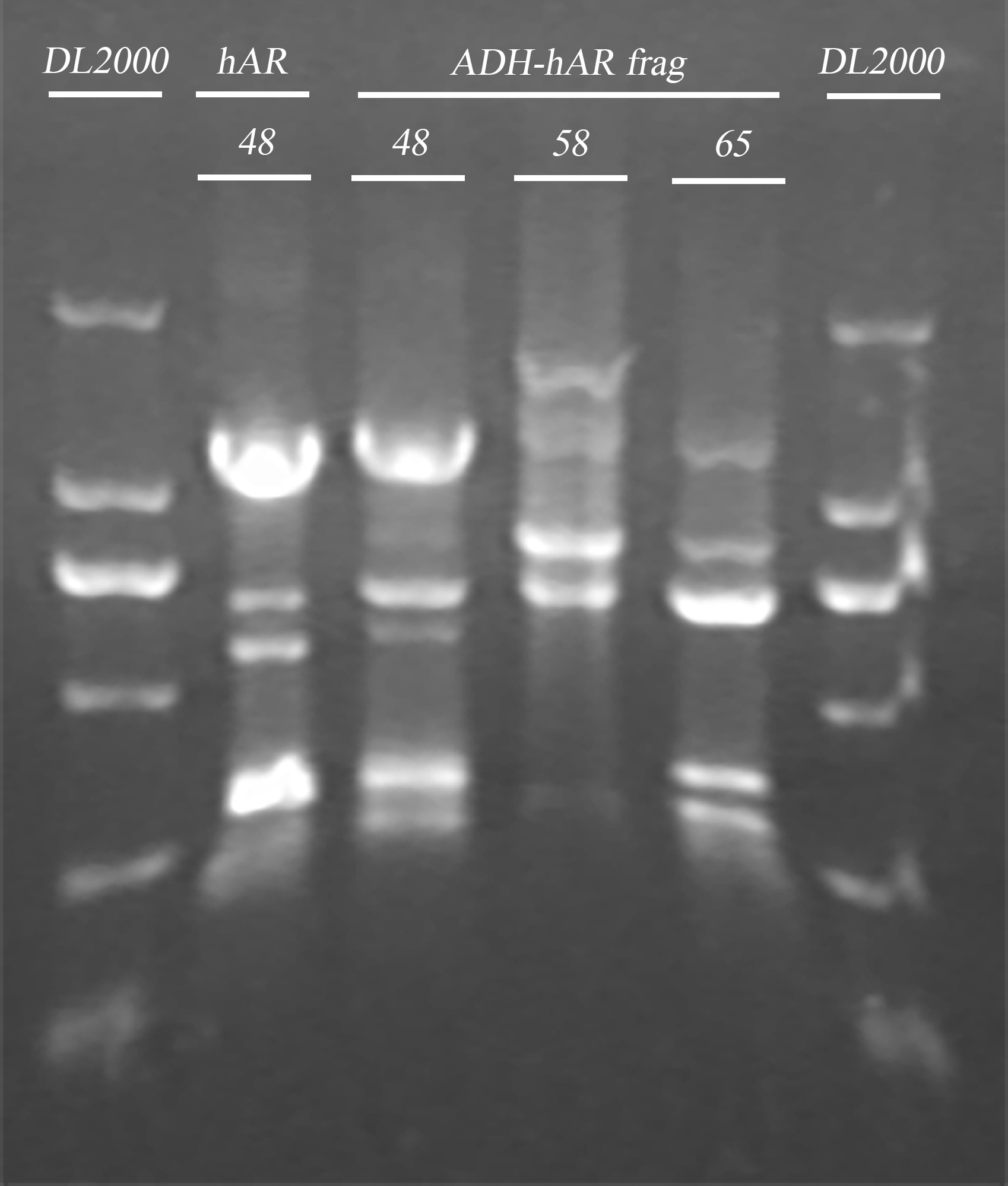
- ADRB2 gene fragment PCR
| Name | Primer | Tm | Template(μl) | ddH2O (μl) | Total(μl) |
| hAR | hAR-R/hAR-F | 48℃ | (48℃) 2.4 | 11.2 | 20 |
| ADH-hAR | TRP-ADH-hAR-R/ TRP-ADH-hAR-F | 48℃ | (48℃) 6 | 28 | 50 |
| ADH-hAR | 58℃ | (58℃) 3 | 31 | 50 | |
| ADH-hAR | 65℃ | (48℃) 6 | 28 | 50 |
Cycle:
| Name | Temperature | Duration | |
| Pre-denaturation | 95℃ | 3 min | |
| Denaturation | 95℃ | 40s | 39x |
| Anealing | 48/58/65℃ | 30s | |
| Extension | 68℃ | 1min 30s | |
| Final extension | 72℃ | 10min | |
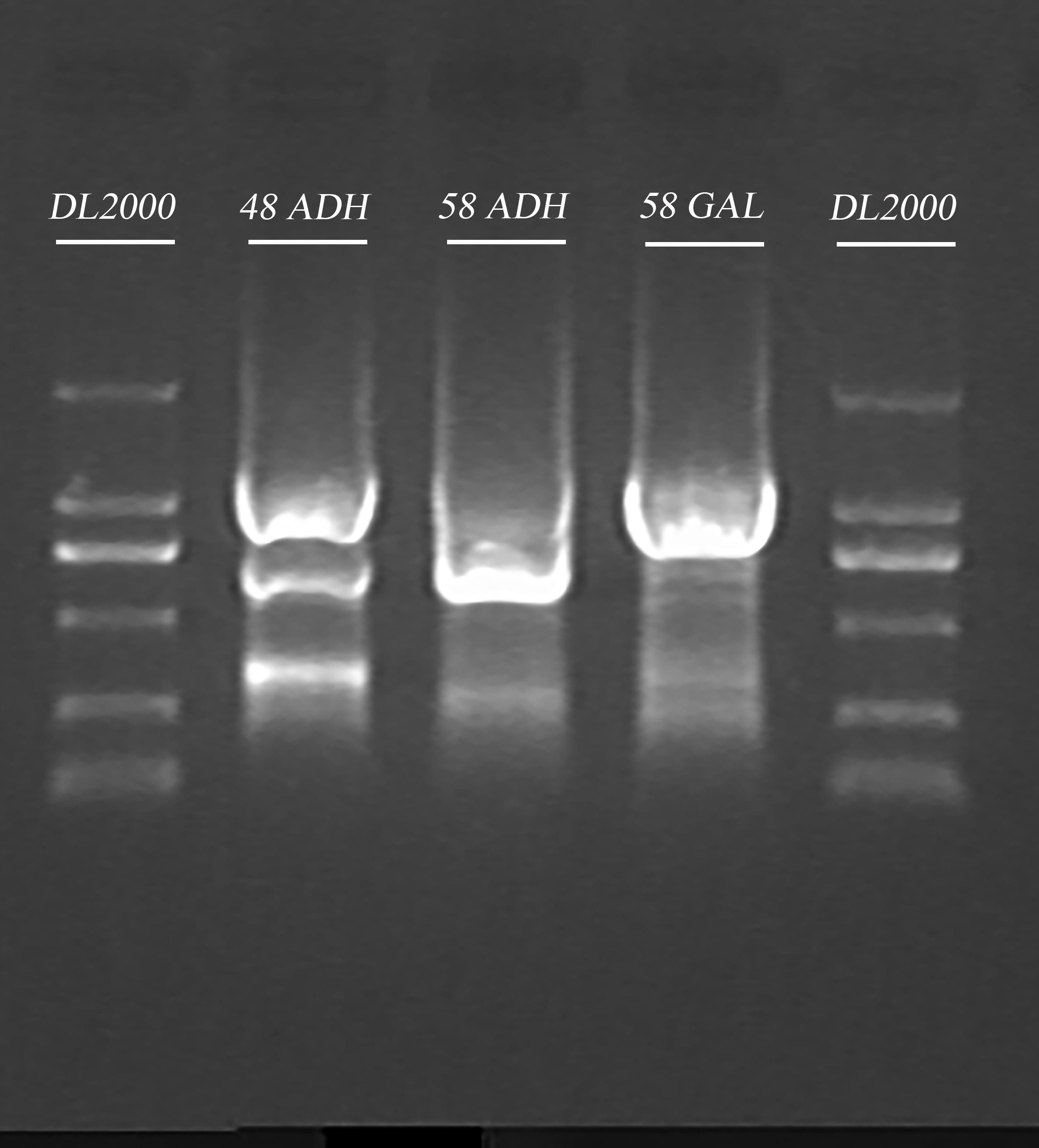
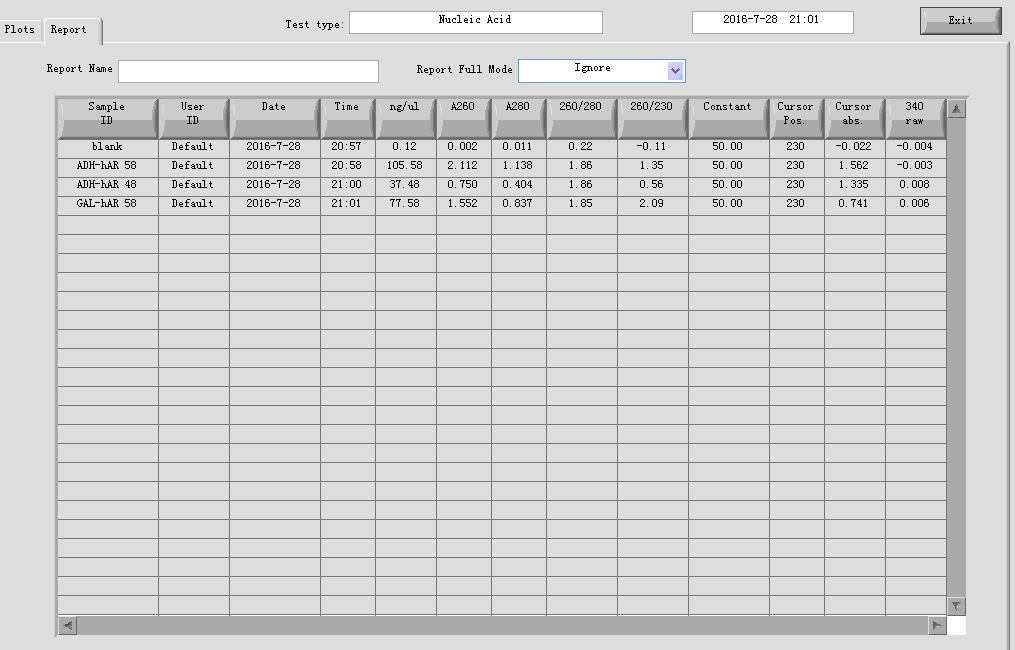
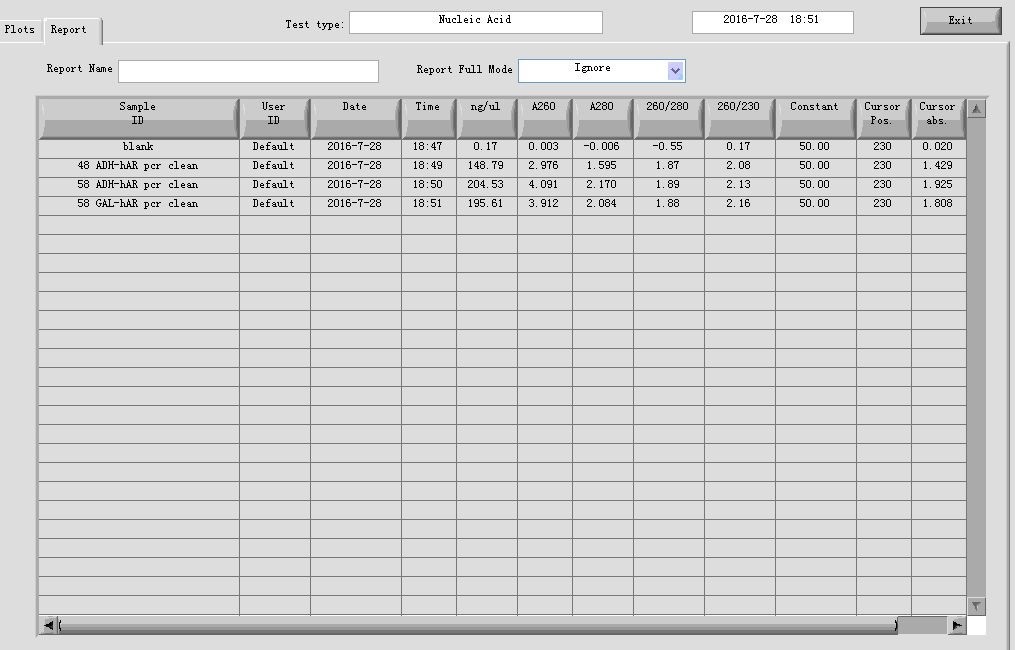
- PCR cleaning
Mixed the parallel groups in step 1, then added equal volume of Buffer CP to them, and follow the standardized protocol provided by the manufacturer. The final elution volume is 30 μl.
- Gel recovery
Ran the samples obtained in Step.3 and recovered the gel. The elution volume is 30 μl.
| Gel | DNA sample volume (after PCR cleaning) | 6 x Loading buffer | DL2000 |
| 1% agarose | 30 μl | 6 μl | 10 μl |
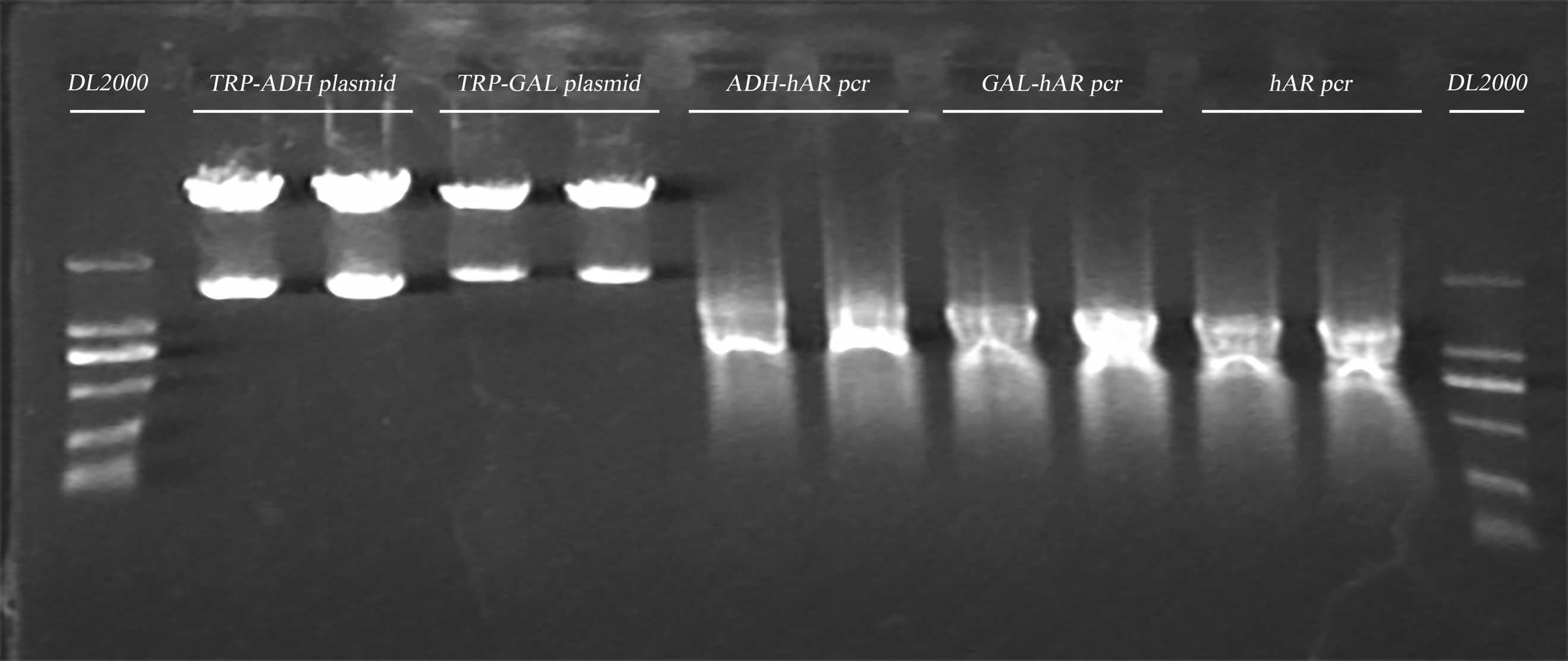
- Insert the fragment into the plasmid TRP-ADH/TRP-GAL
The ligation was achieved using ClonExpress II One Step Cloning Kit following the manufacturer’s protocol.
- Transformation of DH5a competent cells
| DNA volume | Competent cells | LB media | Spread on plate |
| 10 μl | 100 μl | 600 μl | 350 μl |